Table of Contents (click to expand)
Scientists experimented on and observed elements and their behavior, which helped them figure out the existence of atoms and frame the atomic theory.
The atomic theory came into being long before the first transmission electron microscope, which means that we knew about atoms long before we saw them!
Who ever said you need to see things to know about them! We haven’t seen gravity, or electricity, or even magnetism and yet we know a great deal about these things. Science has always been kind of weird in the sense that things known to us are not solely available through visual observation. Curiosity drives all scientific breakthroughs, but when people lacked enough physical and tangible proof, they prove their theories through indirect results. This method of determining results is the foundation of the modern atomic theory.
Recommended Video for you:
Why Can’t We See Atoms?
Quite simply, because they’re so incredibly small! An object is visible when it deflects the light falling on it. The size of atoms falls between 30-300pm, which is approximately of the order 10-12m. For optical microscopes, atoms are invisible, i.e., atoms do not interact with the light particles, so there is no deflection. It wasn’t until the invention of electron microscopes that we first got a glimpse of the atom. An electron beam, which has a lower wavelength than visible light, is scattered when it hits the target; this scattering allows for the creation of an image. There are many more advanced microscopes that not only allow us to observe atoms, but also aid in moving atoms around in a sample to study them!
How Did We Think Up The Existence Of An Atom?
Atomic theory formulation spans many years. Here is a rudimentary attempt to timeline the process of atomic theory formulation, which started in the pre-Socratic period.
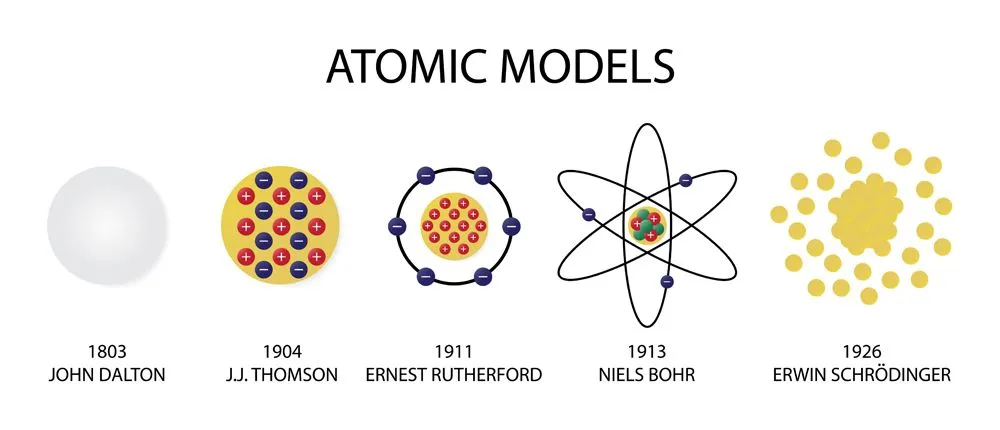
- Leucippus and his student Democritus, the Greek philosophers, were among the first to think of atoms. If a piece of matter is divided into smaller and smaller parts, then all the particles still possess the same properties. If we go on dividing matter, there comes a time when we cannot divide it further. This indivisible particle is ‘atomos’. Democritus believed that atoms were all made of the same basic substance, but differed in shape, size, and arrangement—properties that gave rise to the different materials we observe.
- The atomist teachings were lost for approximately two millennia until the 1800s, when John Dalton first proposed an atomic theory. While trying to figure out why elements only combined in specific ‘whole number ratios’ (like 1:2 or 3:4 etc.), Dalton determined that there must be an indivisible solid, mass-bearing and indivisible particle that is unique for each element. He believed that because this tiny particle was indivisible, compounds could not combine in fractional ratios.
- J.J. Thomson’s ‘plum pudding model’ in the late 19th century was the first model to break the myth of the atom being a solid particle. The cathode ray tube experiment that first discovered electrons led to a modification of the atomic model. The new model was not a solid ball, but had negative charge floating in a sea of positive charge. (It was a positively charged sea because atom as a whole were known to be neutral)
- Ernest Rutherford’s gold foil experiment further acknowledged the fact that the positive charge was only contained in a small part of the atom. Because most alpha rays passed without deflection, the atom had to be largely empty; the few rays that got deflected had likely hit the nucleus. The plum pudding model was replaced by Rutherford’s “nuclear model”, but the position of electrons was still disputed.
- The early 20th century saw the rise of quantum mechanics. Max Planck and Einstein, the pioneers of quantum mechanics, explained that anything that is quantized is allowed to take up only specific values. Niels Bohr, a Danish scientist strongly believed that the structure of an atom was similar to the planetary model. Bohr used the theory of quantization to explain how electrons stay in their orbits, despite orbiting the nucleus and thus not falling into the nucleus.
- Louis de Broglie’s discovery of the dual particle and wave behavior of electrons in 1924 laid the groundwork for a new understanding of atomic structure. Building on this, Erwin Schrödinger developed his famous wave equation in 1926, which describes electrons not as particles in fixed orbits, but as probability waves. We now have a quantum mechanical model of the atom that calculates the probability of finding an electron in a given region. It contradicts Bohr’s assumption of electrons following specific circular orbits, replacing them with fuzzy probability clouds called orbitals.

- While the model may seem complete, the mass of the nucleus was still a mystery. Although we knew about protons and electrons, scientists found that the nucleus weighed more than the combined weight of all the protons—almost twice as much! The discovery of neutrons (whose mass is very similar to protons) in 1932 by Chadwick helped to complete the modern atomic model. The atomic mass of the nucleus was now justified by the presence of these newly discovered neutrons.
As you can see, the modern atomic model is the result of many different observations, questions and experiments. If you observe the way the model has evolved over the years, it becomes clear that due to the lack of visual data available for scientists to analyze, they largely relied on experimental evidence. Remember, this was all way before the first transmission electron microscope first came into being!
Are There Any Other Subatomic Particles?
Modern microscopes, like electron beam microscopes and scanning probe microscopes, have helped us observe the structure of atoms and nano-particles… but there’s more!
Scientists at the Stanford Linear Accelerator Center (SLAC) built a two-mile-long electron accelerator designed to probe the interior of atomic nuclei. Beams of electrons were accelerated to energies of 20 billion electron volts. When such a high-energy beam was targeted at liquid hydrogen and deuterium, researchers observed that electrons began scattering at wider angles and more frequently than anticipated. By the early 1970s, it was realized that there are three scattering centers within each proton and neutron that cause the scattering pattern. This discovery was the first direct evidence of the existence of quarks! Jerome Friedman, Henry Kendall, and Richard Taylor were awarded the 1990 Nobel Prize in Physics for this groundbreaking work.
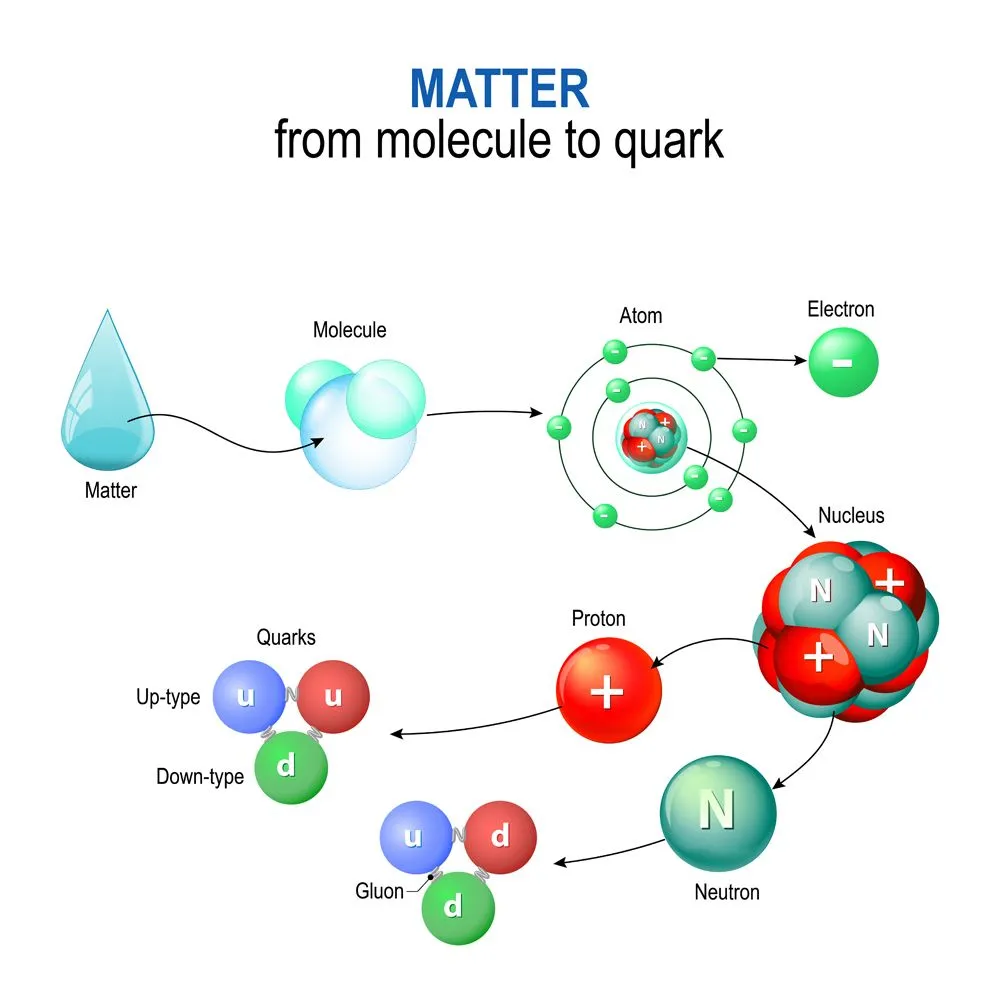
For decades, scientists thought that electrons, protons, and neutrons were the most fundamental subatomic particles, meaning that they were indivisible. However, quarks are the actual elementary particles that make up protons and neutrons! Each proton consists of two up quarks and one down quark, while each neutron consists of one up quark and two down quarks. Electrons, as far as current experiments can tell, remain truly indivisible.
The discovery of the atom and the subsequent discovery of subatomic particles proves the importance of observation and experimentation. The early 20th century did not have powerful microscopes to provide much-needed visual reference, and yet scientists were able to study atoms! Today, techniques like scanning tunneling microscopy (STM) and atomic force microscopy (AFM) allow us to routinely image and even manipulate individual atoms. In 2025, MIT physicists captured the first-ever images of individual "free-range" atoms interacting freely in space, while electron ptychography achieved a record resolution of just 15 picometers—smaller than the atoms themselves. As technology continues to advance, we move ever deeper into the quantum realm, revealing nature's secrets at the smallest scales.
References (click to expand)
- Democritus - Biography & Facts. Britannica
- Questions and Answers - If there is no way in the world to see .... Thomas Jefferson National Accelerator Facility
- Quarks - Hyperphysics. Georgia State University
- Baggott J. E. (2017). Mass: The Quest to Understand Matter from Greek Atoms to Quantum Fields. Oxford University Press
- Seeing atoms - Science Learning Hub. sciencelearn.org.nz