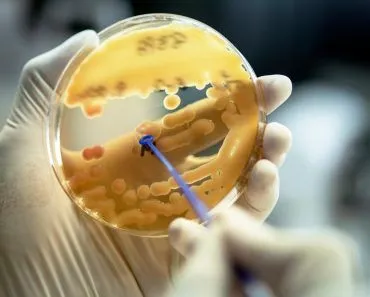
The Limulus Amebocyte Lysate (LAL) test is used to check the sterility of medical equipment. The LAL is extracted from horseshoe crabs. The lysate turns solid in the presence of certain bacterial chemicals that can cause infections.
Have you ever wondered how pharmaceutical companies ensure that all of their products, be it vaccines or syringes, are 100% free from any contamination?
Think about it… there are millions of microscopic organisms all around us at any given moment. If some of them were to hitch a ride on a syringe and enter our bloodstream, things could go sideways pretty fast!

Therefore, to prevent infections, all injectable health products such as syringes, IV fluids, vaccines, etc., must be sterile. The Limulus Amebocyte Lysate (LAL) test is used to check that level of sterility by ensuring that the products are pyrogen-free.
Recommended Video for you:
What Is Limulus Amebocyte Lysate?
Horseshoe crabs or Limululus polyphemus are actually living reservoirs of this key ingredient. The animals are ‘living fossils’, having roamed the Earth relatively unchanged for about 445 million years!
That’s not the only intriguing fact about these mysterious creatures… they’ve also got baby blue-colored blood!

Their blood is blue for the same reason that ours is red. Our blood cells are red in color due to the iron present in the oxygen-carrying molecule, hemoglobin. Horseshoe crabs contain a different oxygen-carrying molecule called hemocyanin. Hemocyanin contains copper instead of iron and therefore imparts a light blue color to the blood cells.
That being said, the most important feature of their blood isn’t the hemocyanin and its blue color; rather, it is the specialized immune cells called amebocytes.
These cells were first discovered in 1956 by Frederick Bang, who found that these cells were highly sensitive to invading bacteria. They attached to the bacteria and immobilized it by forming a viscous gel around it. This protected the rest of the horseshoe crab’s body from harmful bacterial toxins.
This is an important adaptation for a sea-dwelling animal with an open circulatory system like the horseshoe crab. Having an open circulatory system means that their blood is not enclosed in vessels at all times. Instead, it is pumped directly into the body cavity where it freely diffuses in and out of tissues.
Thus, a single bacterium can quickly travel to every tissue in the body and wreak havoc. Since the sea is literally swarming with both harmless and pathogenic bacteria, this adaptation is essential for horseshoe crabs and their longevity.
Additionally, humans found a way to capitalize on this marvelous defense mechanism.
How Does The LAL Test Detect The Sterility Of Medical Equipment?
Pathogenic (disease-causing) bacteria are usually gram-negative bacteria (a type of bacteria). Many of these bacteria contain a pyrogen, that is, a substance produced by the bacteria that causes a fever. One such pyrogen is an endotoxin.
The bacterial endotoxin remains behind even after the bacteria die. If enough endotoxin manages to get into our bloodstream or spinal fluid, we can suffer from fever, shock and organ failure. In extreme cases, it may even lead to death.
At the time of Bang’s discovery, rabbits were being used (read: sacrificed) to check for bacterial endotoxins. Companies maintained large rabbit colonies and inserted a sample into them before moving on to humans.
Rabbits react to endotoxins the same way we do; they develop a fever. The test requires personnel to check the rabbit’s temperature for signs of fever every half hour for a span of three hours, meaning that the method was both time-consuming and expensive (yes, rabbits are high maintenance).

Around a decade later, Bang and a pathologist, Jack Levin, figured out how to extract amebocytes from horseshoe crabs. This was good news for rabbits, but not great for horseshoe crabs.
Limulus refers to Limulus polyphemus, the crab species native to the Atlantic coast of North America that was used for the extraction. Amebocyte refers to the blood cells of the crab. Finally, lysate is the material that is freed by ‘lysing’ or breaking the cells. This lysate is extremely sensitive to bacterial endotoxin.
The Food and Drug Administration (FDA) approved the use of LAL test kits by 1977. Pharmaceutical companies could then simply add LAL to whatever they were testing and turn the vial upside down to see if the solution had solidified.
Since this was much faster and more convenient, it was unsurprising that LAL turned into a multi-million dollar industry.
How Adoption Of LAL Test Kit Affected Horseshoe Crabs Population?
The required lysate is extracted from the blood of horseshoe crabs in a non-lethal way and the horseshoe crabs are then returned to the water within 24 hours.
The pharmaceutical industry has claimed that only 3% of crabs that were bled ended up dying as a result. However, by the early 2000s, it became clear that around 30% of the bled crabs ultimately died. Fewer and fewer horseshoe crabs came ashore during mating season. A research study found that this was because the bleeding process made the animals more lethargic.
So, even if bleeding a female horseshoe crab didn’t kill it, it may make them less likely to mate, which ultimately leads to dwindling population numbers.

Currently, there are only four living species of horseshoe crabs. Limulus polyphemus is on the eastern coast of North and Central America, along with three remaining species on the coasts of Japan, India, and Indonesia. Fortunately, there is still hope for these wonderful creatures.
Alternatives To LAL Test
Biologists from the University of Singapore came up with a synthetic alternative for lysate in the late 1990s. They identified the gene that made a molecule called factor C. This factor C is the specific molecule in LAL that detects bacterial toxins. The biologists inserted this gene into insect gut cells, turning them into mini-factories that produce the genetically engineered protein—Recombinant Factor C (rFC).
In 2016, rFC was approved as an alternative bacterial toxin test in Europe and was also used by some American pharmaceutical companies. However, a major setback came in June 2020 when the American Pharmacopeia—the regulatory body that sets the standards for drugs and other products in the U.S.—refused to accept rFC as an equal to LAL, claiming that its safety was still unproven.
But, other governments, including those of Japan and China, have approved the rFC test.
At the end of the day, the big winners are humans, as usual. Vaccines protect us from all kinds of diseases, including measles, mumps and most recently, COVID-19. However, it is the Limulus amebocyte lysate extracted from horseshoe crabs that prevents infection via drugs, vaccines and other medical products through bacterial contamination.