Table of Contents (click to expand)
Cotton candy is composed of thin strands of glassy sugar fibers. The bottom line is that cotton candy is fiberglass, but fiberglass is not cotton candy.
“I ate fiberglass insulation. It wasn’t like cotton candy like the man said…my tummy itches.”
This is one of the many memorable lines said by Steve Carell in the movie Anchorman.
Swallowing glass fiber like its cotton candy will certainly upset the stomach, but that doesn’t mean they aren’t essentially the same. On a molecular level, apart from their optical similarity, they have quite a bit in common. And, not only cotton candy, there are other types of sugar that act just like glass.
But…
Recommended Video for you:
What Is Glass?
This sounds like a silly question, as we all know what glass is, right? It’s transparent, breaks easily, and is used to make a wide range of objects, from windows to spectacles. However, for scientists, “glass” is just another category of substances, and the title is not held exclusively by the kind made from rock flour.
Glass is a class of amorphous solids with the irregular molecular arrangement of a liquid and the rigidity of a solid crystal. These substances are formed when a viscous liquid cools down quickly and doesn’t have enough time to arrange the molecules in an orderly fashion, as crystals typically are. This means the term “crystal glass” is actually an oxymoron.
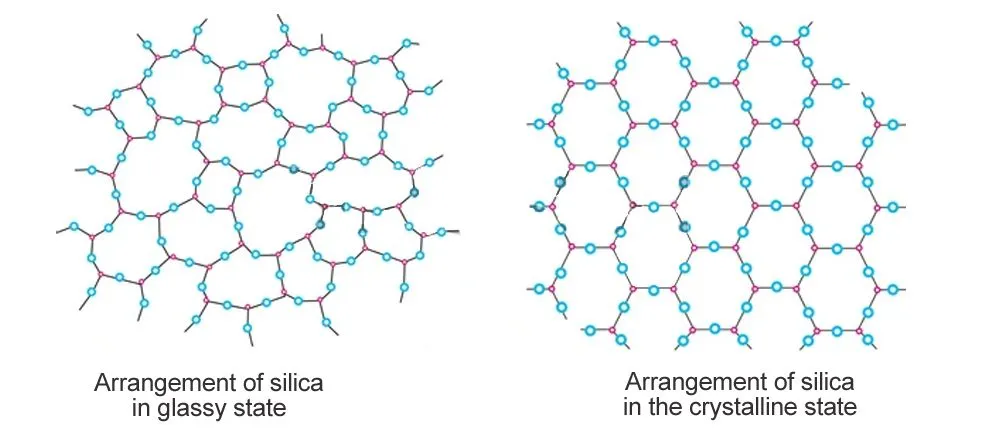
Glass is also not always brittle; at times, it’s a few hundred times stronger than steel. Scientists have discovered that when molten metal alloys are supercooled at thousands of Kelvin per second, they form something called metallic glass, a substance as strong as metal and as versatile as plastic.
You’re not so sure about the real definition of glass anymore, are you? Well, neither are scientists.
Some claim that glass is a non-crystalline solid, whereas others argue that it should be classified as a frozen liquid; the answer to what is glass? is crazier than M. Night Shyamalan’s film of the same name.
Speaking of films, imagine a fight scene in which a villain hits the protagonist on the head with a glass bottle. Seems painful!
Well, the props go to good acting and not to the prop glass bottle. The bottle that landed on the head of the good guy was indeed glass, but not glass like mirrors or crockery, but the kind that might stick to your molars. Because in fact, it’s just hard candy, a.k.a sugar glass.

From Sugar To Glass
Granulated sugar is made of a carbohydrate called sucrose. Furthermore, sucrose molecules love to form regular 3D structures, which is why sugar is crystalline. When dissolved in water, these molecules break apart from each other and attach themselves to water molecules.
Even though sugar is highly soluble in water, it reaches a point where no more sugar can be dissolved. In this supersaturated state, the sugar starts to recrystallize. Heating the supersaturated sugar stops the recrystallization and results in sugar syrup.
Sugar syrup is no less than a shapeshifter, and mankind has mastered the art of manipulating a simple syrup (along with some additives) into a wide range of magical treats. Be it fluffy marshmallows, chewy toffee, or rock candy… everything begins with simple sugar syrup and a trusty candy thermometer. To read more about candy making, click here.

Glass is obtained when a mixture of silica/sand, soda, and limestone is melted in a furnace at 1700⁰C and rapidly cooled. Candy glass is made from a solidified mix of sugar, water corn syrup, and cream of tartar. The three keys elements to making perfect hard candy are the water content, temperature, and the rate of cooling for the molten mixture.
To attain a hard crack state, we need a highly viscous liquid. Thus, the water content in the mixture is boiled off, until the mixture is 97-99% sugar. The syrup is carefully heated until reaching the hard crack temperature (145°–152°C). If overcooked, the mixture will turn into a sticky caramel. The syrup is then cooled down right below its glass transition temperature. The mixture is cooled rapidly to prevent the sucrose molecules from forming an ordered network and crystallizing.
After all these steps are followed carefully, we end up with an amorphous, soft solid sheet of candy that is also a form of glass.
Is Cotton Candy Similar To Fiberglass?
A cotton candy machine has a heater that melts sugar to a hard crack stage. The melt is then extruded through hundreds of fine nozzles. The molten liquid solidifies as soon as it touches the outside air. Since the sucrose molecules do not have time to arrange as crystals, they freeze into a glassy state. The spinning motion of the machine creates a centrifugal force that pulls the melt into fine sugar glass fibers. Fiberglass, which is made from molten silica, is manufactured in a large industry-grade machine whose working principle is very similar to a cotton candy maker.
The bottom line is that cotton candy is fiberglass, but fiberglass is not cotton candy!
References (click to expand)
- T Husband. The Sweet Science of Candymaking. The American Chemical Society
- A Note on the Scientist's Definition of Glass. cmog.org
- Hartel R. W.,& Hartel A. (2014). Candy Bites: The Science of Sweets. Springer New York
- Metallic Glass | MATSE 81: Materials In Today's World. The Pennsylvania State University
- Science and Property of Sugar Glass - Lehigh University. Lehigh University












