Table of Contents (click to expand)
Bose-Einstein Condensate is a state of matter only observed for bosons around 0 Kelvin, wherein the particles bunch up and behave as one.
If you are reading this, you might have, at some point in your life, heard or read about something called a ‘Bose-Einstein Condensate’ (also called BEC). You might be buzzing with questions, but don’t know where to begin. If so, then you’ve come to the right place to learn everything about BEC, but in a beginner-friendly way!
Let’s begin by stating two obvious facts about Bose-Einstein condensates. First, it is categorized among different states of matter, so we know that it is a state of matter. However, it is not as common as others, because if it were, we would have learned about it in primary schools along with solids, liquids, and gases.
This brings us to the second fact. This condensate is not naturally available. It must be synthesized in laboratories that can provide the appropriate conditions for it to be sustained. Or we must look for them in delicately extreme conditions, such as the inside of neutron stars, to find possible examples of this state.
You are probably wondering… how did someone even think of the existence of such a material when it hadn’t ever been seen in nature? How do they make this Bose-Einstein Condensate in labs, and what makes it so special that it has its own category of matter? Well, let’s dive right in and get the answers to all those questions, starting with the origin itself!
Recommended Video for you:
Story Of BEC
Almost a century ago, Satyendra Nath Bose sent his unpublished work on the quantum statistics of light quanta to Einstein. He basically gave a statistical study or theory of light particles and their quantum nature. Einstein was impressed by his work and submitted it for publication on behalf of Bose. Einstein furthered Bose’s idea and from it came the concept of Bose-Einstein statistics.

Basically, Bose-Einstein Statistics deals with particles having integer spins (0, 1, 2, 3,…), which we now call Bosons. As the name suggests, the basis of Bose-Einstein Statistics is the study of the statistical distribution of bosons and their resulting behaviors. We now know that there is another category of matter particles called Fermions, which have half-integer spins (1/2, 3/2, 5/2,…). Fermions obey their own quantum statistics theory, called Fermi-Dirac Statistics.
It was through his work that Einstein realized one consequence of a property of Bosons. The property itself was that bosons with the same quantum numbers can occupy the same quantum states. These states include properties such as momentum, energy, etc.
The consequence was the concept of BEC, but what is the meaning of the property and why is it so remarkable?
What Makes It So Special?
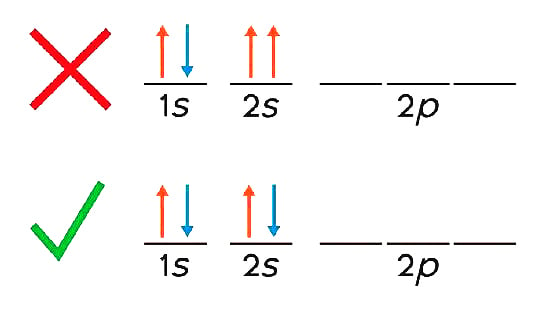
Think of an atom. Imagine that there is a vacancy in an energy level (the 2s level, for simplicity) and two electrons can occupy that vacancy.
Now remember, electrons have many quantum numbers that define their quantum states, such as the principal quantum number, angular quantum number, magnetic quantum number, etc. However, for the 2s level, the two energy levels only differ by the spin quantum number (+1/2 and -1/2 spins).
Let’s say that an electron with spin-up (+1/2) occupied one position, as shown in the diagram. Now, the room available for another electron can only be occupied by a spin-down (-1/2) electron, such that the overall configuration of quantum states is different.
This property of excluding any particle with the same quantum numbers from occupying the same energy level is called Pauli’s Exclusion Principle.
Now, let’s look at the electron itself. It has a half-integer spin (1/2), meaning that it follows Fermi-Dirac Statistics. But that is not the case for bosons! Bosons with the same quantum numbers can exist together at the same energy level, meaning that they do not obey Pauli’s exclusion principle. So what happens when bosons come together and occupy the same energy level at the same time… and how does it happen?
Einstein imagined that if a system of bosons is cooled enough so that their random motions are eliminated, they might all come together to occupy the lowest quantum state. This is when the quantum nature of the particles would be magnified.
You should know that when particles share the same energy level, they all have the same energy. At that moment, we can still distinguish between particles because of their quantum numbers, such as spins, as seen before. However, in this system of bosons, where they can occupy the same quantum states, including the same energy levels, there is nothing differentiating each particle. Basically, all the particles are indistinguishable!
In short, they lose their individual identities and behave as one body, like a superatom!
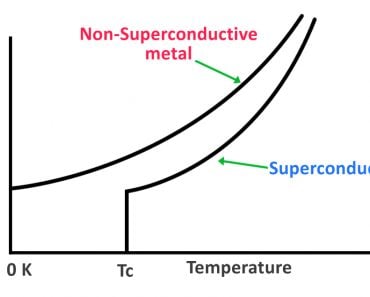
This process of forming this superatom is what Einstein called Bose-Einstein condensation, and the superatom itself was called the Bose-Einstein Condensate. It has very unique properties, such as superfluidity, and in many cases, superconductivity as well! However, it’s the behavior of this large group of particles coalescing and acting as one huge particle (magnifying its quantum nature) that makes it so interesting to study.
Making Of BEC
The question remains, where do we find this peculiar state of matter? The answer is… wherever we might be able to maintain a temperature of approximately 0 K. The bad news is, we have never reached absolute zero in a lab. Moreover, maintaining a temperature even close to absolute zero is a fairly difficult task!
But what about in space? After all, we all know that space is really cold. Unfortunately, the reality is that we have not discovered any location in the visible universe at 0 K. Even the average background temperature of space is around 2.725 K.
Einstein himself was skeptical of the production of BEC. How would anyone be able to achieve the conditions appropriate for it to happen? He called this a paradox and left it at that.
Almost 70 years later, that paradox was made possible at last!
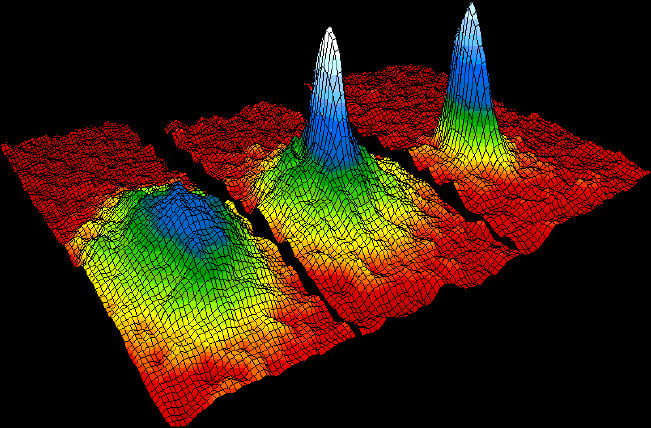
The first BEC was made by Eric Cornell and Carl Wieman’s team at JILA, composed of Rubidium atoms. This was only possible because they could achieve temperatures in the nano-Kelvin range, which was a necessity for Bose-Einstein condensation to set in. Marvelously, they were able to go down to as low as 20nK! Even before reaching that temperature, they had begun to observe the condensation process setting in at 170nK! Just a few months later, Wolfgang Ketterle and his team were able to observe the phenomenon in Sodium atoms. This monumental achievement led Cornell, Wieman, and Ketterle to share the Nobel Prize in Physics in 2001.

Now, the process of their formation isn’t as simple as just cooling them to incredibly low temperatures (which is also very difficult!). The full process involves magnets and lasers to trap the particles and cool them. These particles also need to have low density, such that they don’t immediately condense into solids or liquids instead.
And even when all these meticulous variables are achieved, the BEC forms for only a short period of time!
Conclusion
Now that we understand the origin, basics, and important properties of BEC and its formation, we can conclude by answering the obvious question. What is it useful for?
Thus far, it has helped us understand quantum many-body phenomena, as well as in the study of exotic phenomena, such as the emission of Hawking radiation! In other words, the study of Bose-Einstein Condensate isn’t just limited to the quantum world, but it stretches into the cosmos and beyond!