Table of Contents (click to expand)
A world without salt water would be a boon to humanity, but it would also decimate marine life and affect our global temperature and weather dramatically, making human life on Earth incredibly complicated.
What if we didn’t have to worry about global water consumption? What if you could simply shower as much as you wanted? What if being marooned in the middle of the ocean didn’t kill you of thirst? What if a trip to the beach and a trip to the well were one and the same?

A world without salt water would indeed be a boon to humanity, but what would it cost?
Recommended Video for you:
The Big Question
To begin with, why are seas and oceans salty?
Well, they weren’t always like that. Around 3.8 billion years ago, when the Earth’s surface had finally cooled to the point where water vapour had turned to liquid, there was nothing but the sparkling, unadulterated, purest form of water you could imagine.
Yes, a really, really long time ago, all of the water on Earth was freshwater!
However, that didn’t last long… the universe imposes its will sooner or later. The climatic cycles began soon after the planet cooled. Whenever it rained, carbon dioxide from the atmosphere dissolved into the falling water, making the rain slightly acidic. As it rained, it caused rocks to erode. From there, rainwater slid towards nearby rivers and streams, carrying along both dissolved and undissolved loose salt and minerals. These runoffs ended up in larger water bodies—seas and oceans.
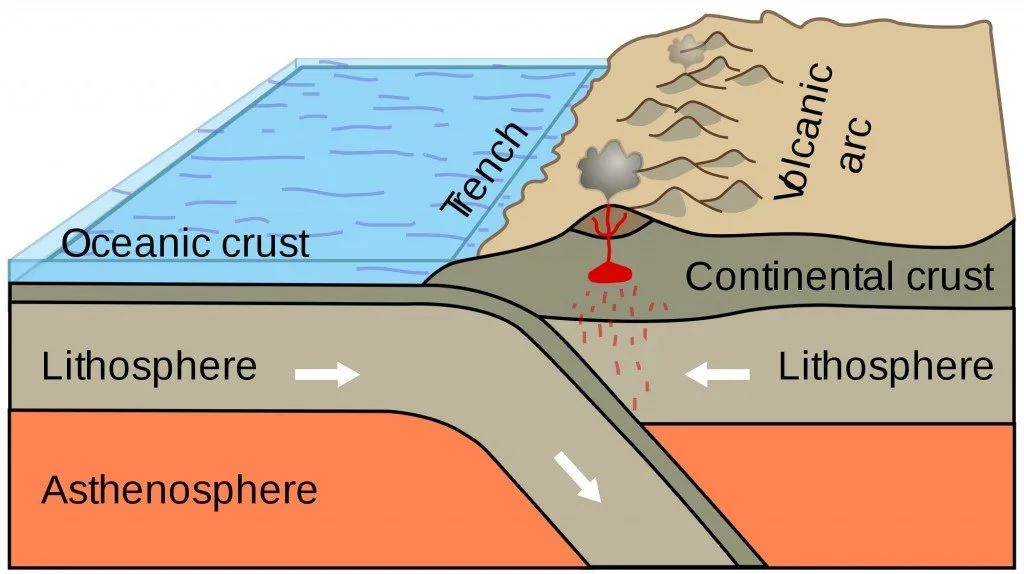
Adding to that, more salt and minerals were expelled from hydrothermal vents and submarine volcanoes. Now, consider that this whole process has been consistently occurring for more than 3.8 billion years! That’s quite a lot of salt, as you can imagine.
In fact, the salt in the oceans is so abundant that when spread across all the landmass on Earth, it would create a single towering layer of salt, 40 stories tall!
No wonder 97% of the world waters are saline…
Freshwater Oceans: Boon Or Curse?
At our current rate of water consumption, the demand for freshwater doubles every 20 years around the world. This will lead to one-third of the world being deprived of freshwater by 2025. With statistics like those, the idea of freshwater oceans doesn’t sound like a privilege so much as a desperate necessity.
However, an ocean without salt would decimate marine life and affect our global temperature and weather dramatically, making human life on Earth incredibly complicated.
Marine life
There are roughly 230,000 known marine species in the ocean and an estimated 2 million more that have yet to be discovered, but if the sea were to become desalinated, we’d likely never get the chance to find those new forms of life.
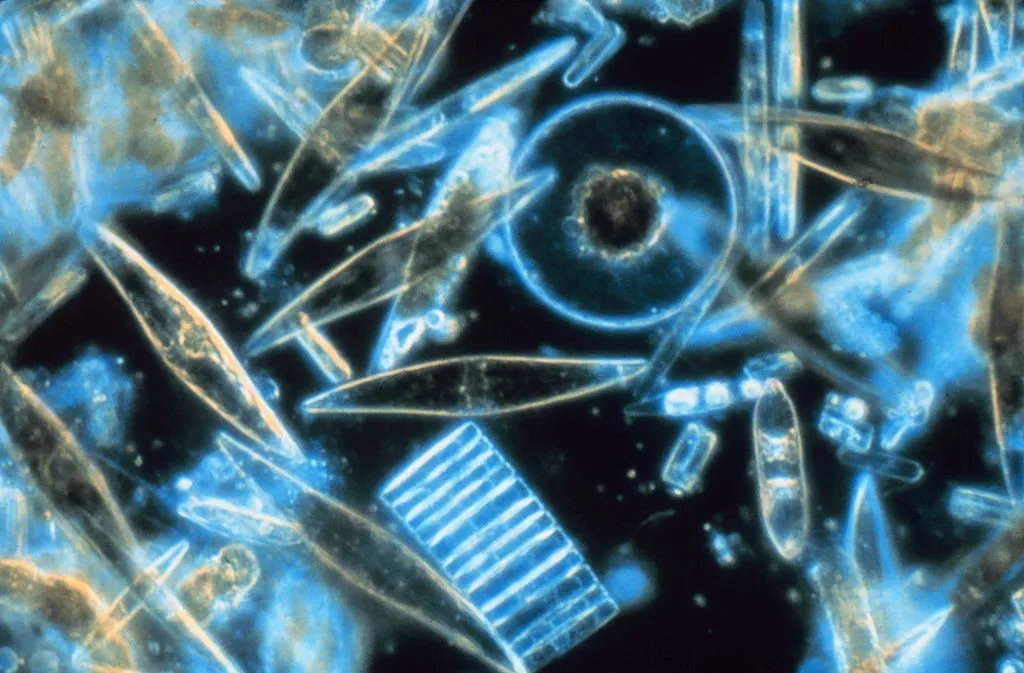
The marine organisms that would be affected most are plankton and phytoplankton, which form the base of all marine life. Saltwater fish have evolved and adapted to the saline conditions, drinking salt water to stay hydrated and getting rid of excess salt by pumping it out. Other fish, such as salmon, have adapted to tolerate both freshwater and saltwater, but they are “one in a thousand” species with that ability. On a massive scale, all saltwater species would perish.
Plant Life
Along with marine animals, marine plant life would also be affected. Underwater algae account for almost half the photosynthesis occurring on this planet!

Photosynthesis plays a crucial role in supplying our planet with food and oxygen, namely by converting carbon dioxide from the atmosphere into the essential oxygen required for breathing. Hence, without algae, not only would we get less oxygen, but we would also have a lot more carbon dioxide in our atmosphere!
With significantly less photosynthesis and extremely volatile climatic conditions, our world would no longer support the diverse plant life it has now. The food chain itself would collapse. Most species, including humans, would not survive for long.
World Weather
This escalation of the greenhouse effect would make some parts of the world unbearably hot.
This would be most noticeable at the Equator since our ocean currents would no longer circulate warm water and air currents the way they used to.
Convection currents move warm waters from the Equator to the farther north, while colder waters from the north cool the hotter areas down south. In the Equatorial region, warmer water is able to carry more salt (the solubility of solvents increases with temperature), causing this denser water to sink to lower depths, while cooler water flows over the top. In the far north, the water cools enough to freeze and form sea ice. As the water freezes, salt is left behind, and naturally, this makes colder water up north much denser, allowing it to sink lower, which makes way for the incoming warmer waters from the South.

Without salt, this whole balanced and intricate process breaks down. For starters, hurricanes would become much more frequent—and more deadly! Basically, our weather and climate would be astoundingly different from what we experience now! Luckily, you wouldn’t survive long enough to experience the full wrath of global weather. You’d either freeze at the poles or overheat at the Equator, die in a natural disaster, or starve to death since you’d be living in a world with no plant or marine biology.
Conclusion
It’s quite funny if you think about it… all we desire is a world without a water crisis, where freshwater is as abundant as grains of sand. However, now that you know what the consequences of such a saline shift might be, you might not be so quick to beg for the end of saltwater.

Nature creates every living and non-living entity in a convoluted balance. A mismatch here, an accident there, and you’re doomed to drown in the perils of unbalance. It would be better for all of humankind to focus their resources and intellect on conserving and preserving what little fresh and consumable water we have left.












