Table of Contents (click to expand)
Light is both a particle and a wave. Light has properties of both a particle and an electromagnetic wave but not all the properties of either. It consists of photons that travel in a wave like pattern. The debate has raged for generations amongst the giants of the physics community regarding the nature of light, namely whether it is a particle or an electromagnetic wave.
Light is both a particle and a wave. Light has properties of both a particle and an electromagnetic wave but not all the properties of either. It consists of photons that travel in a wave like pattern.
The debate has raged for generations amongst the giants of the physics community regarding the nature of light, namely whether it is a particle or an electromagnetic wave. For centuries, this mysterious and elusive phenomenon left scientists baffled because with each experiment conducted to define its nature, it seemed to change the way it behaved.
In simple terms, light is one of nature’s freaky exceptions, and is considered to be both a wave and a particle. This variability is also one of the fundamental tenets of the theory of Quantum Mechanics. Let’s look at what happened over the years as people came to this important conclusion.
Recommended Video for you:
Light Is A Particle
The idea that light is a particle was first put forward by Sir Isaac Newton in 1704, in his corpuscular theory of light. Newton argued that properties such as the reflection and refraction of light could be explained if light was made up of tiny particles, or “corpuscles.” His view dominated optics for nearly a century, until 19th century experiments by Thomas Young and others made a strong case for light as a wave. The particle picture was later revived in a modern form by Albert Einstein in 1905, when he proposed that light consists of discrete quanta of energy (photons) to explain the photoelectric effect.

Classical waves do not travel in straight lines and cannot easily exhibit the sharp shadows and predictable reflection that Newton pointed to. So why was light eventually rejected as a pure particle? The partial answer is that it did not have all the properties that define ordinary matter. A particle of matter has properties such as mass and volume. The smallest unit of light is the photon, which has zero rest mass and no volume in the everyday sense. On top of that, experiments by Young, Fresnel and Maxwell during the period between Newton and Einstein revealed clear wave-like properties of light — interference, diffraction and electromagnetic behavior — which led most 19th century physicists to conclude that light was energy in the form of waves, not matter.
Nope, Light Is An Electromagnetic Wave
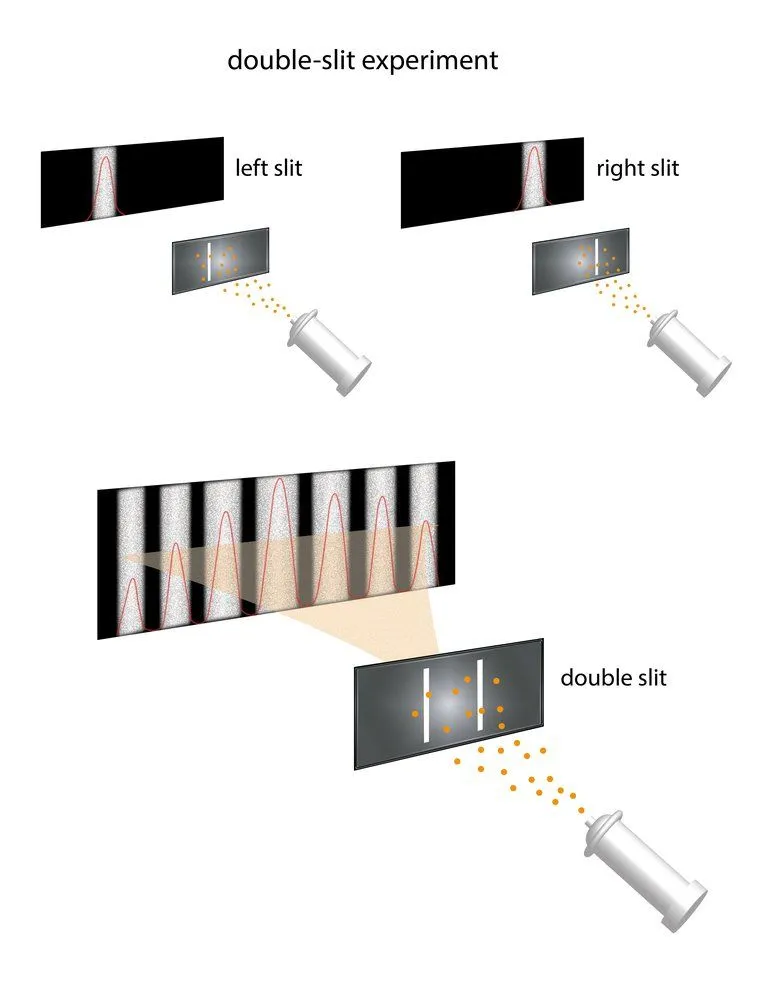
A number of scientists, including Young, Fresnel and Maxwell, are credited with investigating the wave-like properties of light. A wave is a transfer of energy from one point to another without the transfer of material between the two points. In 1801, Thomas Young performed his famous double-slit experiment, which was instrumental in establishing the wave-like properties of light, such as interference and diffraction. He passed a beam of light through two narrow, closely spaced slits and observed the pattern it formed on a screen placed behind the slits.

If the corpuscular theory of light (light is a particle) proposed by Newton were true, then the pattern on the screen should have been just two bright bands, one behind each slit. Instead, the screen showed a series of alternating bright and dark fringes spread out across it, which indicated that light exhibits interference — a property characteristic of waves. Interference is a phenomenon in which two waves either have an additive or subtractive effect on each other’s intensity, making the resultant wave either greater or lower in amplitude.
Einstein And The Photoelectric Effect
In the late 19th century, physicists noticed that when light shines on certain metals, electrons are ejected from the metal surface. The strange thing about the photoelectric effect is that the energy of those ejected electrons (photoelectrons) does not change with the brightness of the light — it only depends on the light’s frequency or color. If light were simply a continuous wave, then more intense light should give the electrons more energy, but that is not what was observed. In 1905, Einstein explained this puzzle.
Einstein proposed that light is actually made up of tiny packets of energy that travel and propagate in a wave-like manner. He called these packets quanta of light; today we call them photons. He argued that when an electron in the metal absorbs a single photon, it takes that photon’s entire energy and is ejected from the surface. The higher the frequency of the photon, the more energy it carries, and the more energetic the ejected electron. This neatly explained why frequency, not intensity, controls the energy of the photoelectrons — and it earned Einstein the 1921 Nobel Prize in Physics. The dual nature of light is illustrated even more vividly through the double-slit experiment.
When light is sent through two narrow, parallel slits, its wave-like nature causes the waves passing through the two slits to interfere with each other, producing bright and dark bands on the screen behind — a result that would not be expected if light consisted of classical particles. And yet, the light is always absorbed at the screen at discrete points, meaning as individual particles (photons), not as a smeared-out wave. Even more surprisingly, when detectors are installed at the slits to track which slit each photon passes through, the interference pattern disappears and each photon behaves like a particle going through just one slit. In 2025, MIT researchers reproduced this very effect at the most fundamental quantum level, using ultracold atoms as the “slits,” and confirmed that the more precisely you pin down which path a photon takes, the more its wave behavior fades — a striking modern confirmation of wave-particle duality.
In other words, light may have Dissociative Identity Disorder (DID), but at least we can stop arguing about it now!











