Table of Contents (click to expand)
The body’s pH level is important for good health. pH is a measure of how acidic or alkaline something is. A pH of 7 is neutral, while anything below 7 is acidic and anything above 7 is alkaline. The body tries to keep its pH level at 7.2-7.4, which is slightly alkaline. There are three ways the body regulates pH: respiration, the kidneys, and the chemical buffers in the body. Our diets and lifestyle choices are the biggest factor in determining our pH level. To maintain a healthy pH, eat fruits and vegetables and drink alkaline water.
Our bodies are incredible machines, but just like any complex structure, it takes a lot of work to make it run smoothly. One of the most basic elements of our health and the wellness of our body is the pH level we maintain. For those who don’t know, pH is a measurement of the acidity and alkalinity within the body. A neutral pH is 7.0. If the number falls below that measurement, the body is said to be acidic, while a number above that is considered alkaline.
Whether your body is acidic or alkaline can have serious ramifications on your health, which leads to the obvious question… what is the ideal pH of the body?

Short Answer: Depending on who you ask, the ideal pH for human health is between 7.2 and 7.4, slightly on the alkaline side, as an acidic environment makes you more vulnerable to disease.
Recommended Video for you:
Why Does pH Matter?
The last time you read about pH, it very well may have been in a high-school science class, but whether you like it or not, pH has an effect on your life every single day. Basically, an imbalance of pH in the body allows diseases and illness to take root, primarily if your body is too acidic. Excess acidity can also damage tissues and compromise the integrity of your organ systems.
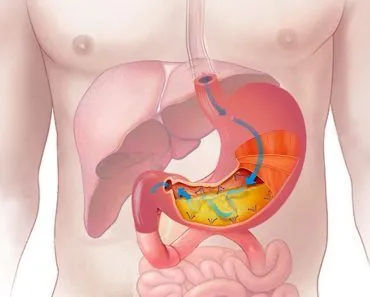
As a basic introduction, for those who slept through most of their science classes, pH ranges from 0 to 14, and a perfectly neutral pH is 7. As an example, the highly acidic conditions in the stomach, which are necessary for the body to digest food and absorb nutrients, is between 2 and 3 – extremely acidic. On the other hand, water that you may drink fresh from a mountain stream – after it has picked up countless extra minerals from rocks, is between 8 and 9 – moderately alkaline.

Everything that we consume, as well as many lifestyle choices and activities, has an effect on the pH of our body, and over the course of a single day, our pH fluctuates; 6 – 8 is the typical range. However, our body constantly works to maintain a stable pH level and ensure our health. Our saliva and urine tend to have a pH of 6.4-6.8, whereas the blood and tissues of the body should remain between 7.2 and 7.4.
How Does The Body Regulate pH?
As it turns out, the body has a number of ways to regulate pH in place, beginning with a chemical buffer called bicarbonate (HCO3) that can be regulated by the kidneys. The H+ ion in bicarbonate binds with hydrogen, creating H20 and CO2, which can be eliminated from the body through urination and exhalation, respectively. This will help to reduce the acidity of the body, edging the total pH closer to a slightly alkaline level. Conversely, when the body is too alkaline, H+ can be released into the blood, making it more acidic.
The second way the body regulates pH is through respiration. When we exhale, the amount of CO2 in the blood (and thus bicarbonate) also decreases. This process can be sped up by rapid breathing in and out, also known as hyperventilation. This isn’t a long-term fix, however, but respiration, in general, is a second way that the body controls the amount of bicarbonate in the body.

Finally, and perhaps most importantly, are the kidneys, which can absorb acidic or basic materials depending on what the body needs. Given that we tend to have more acidic diets in the modern world, our urine is almost always slightly acidic, because the body is trying to excrete those acidic components and keep the pH level at a slightly alkaline level. The lungs, kidneys and chemical buffers in the body work in constant conjunction to maintain a healthy environment in our blood and tissues, which strengthens the immune system and prevents the formation of disease.
How To Maintain A Healthy pH
While the body has certain mechanisms in place to help with the balance of pH in the body, your dietary and lifestyle choices are the ultimate determinant in whether you have a proper pH level. There are many different factors that can cause acidity in the body, including chronic stress, drug and alcohol abuse, exposure to chemicals, pesticides, herbicides, pollutants and radiation, excess animal protein from processed food, and artificial sweeteners, just to name a few.
If you don’t adjust your behavioral patterns and eating habits, you are increasing your risk of inflammation, headaches, fatigue, diabetes, cancer, stroke, arthritis, skin problems, ulcers and unexplained weight gain. An acidic environment makes it harder for the body to function normally, and results in acidosis. One of the dangers of acidosis is that it is hard to detect. Since the body does its best to regulate acidity, when you take a pH test, it will often appear that you have a balanced pH. However, if you have an excess of acid-inducing foods and lifestyle choices, the body will sap minerals from tissues and bones in order to compensate.
Over time, this will significantly increase your risk of osteoporosis and the health issues mentioned above, because proper electrolyte levels (calcium, potassium, magnesium and sodium) are essential for health. Even if a pH test shows that your body is “balanced”, this might be misleading. Scholars believe that chronic low-grade acidosis is extremely common, particularly in Western cultures, and contributes significantly to ill health.
In terms of your diet, fruits and vegetables are the best options to balance your pH, as they are naturally alkaline in nature. Some people may point to citrus fruits as sources of acidity, but in fact, when the body metabolizes citric acid, it has an alkalizing effect, which is a good thing. Another option that many people are turning to is alkaline water, which has a pH between 8 and 9.
As you can see, a few conscious decisions can help your body maintain its pH at a health level around 7.3, which should keep you safe from a wide range of long-term health issues.
References (click to expand)
- Pitts, R. F. (1950, September). Acid-base regulation by the kidneys. The American Journal of Medicine. Elsevier BV.
- Mudge, G. H., & Vislocky, K. (1949, May 1). Electrolyte Changes In Human Striated Muscle In Acidosis And Alkalosis. Journal of Clinical Investigation. American Society for Clinical Investigation.
- Alkaline Diet: Does pH Affect Health and Wellness?. foodandnutrition.org