When water freezes, its molecules get arranged in a crystalline structure, thereby attaining a defined shape. This crystalline structure is less dense, and since there are gaps between individual molecules in the structure, the overall volume increases and water ‘expands’.
From a cursory glance, the phrase “water expands when it freezes” may not make sense, because in liquid form water has no definite shape or form, and therefore seems to occupy more space. Also, when it freezes, water takes on a clearly defined form, which seems quite the opposite of “expansion.”
Let me therefore begin by breaking down the question posed in the title of the article.

Recommended Video for you:
Does Water Really Expand When It Freezes?
Yes, water expands when it freezes. In fact, a given volume of liquid water increases by roughly 9% after it turns into ice. So a 100 mL bottle of water, if frozen completely, would need about 109 mL of space — which is exactly why a sealed bottle of water cracks in the freezer.
Note that the word “expands” here indicates an increase in volume. A technically sound way to put it would be: water’s volume increases when it freezes. You can test this with a simple experiment — if you lower the temperature of water, you will notice that its volume decreases as it becomes more and more “intact”… until you cross a curious threshold.
That threshold is 3.98 °C (about 4 °C), the temperature at which liquid water reaches its maximum density of 1.000 g/cm³. Cool it any further and, instead of contracting like most liquids, it starts to expand. By the time it has fully frozen at 0 °C, ice has a density of only about 0.917 g/cm³ — roughly 9% lower than liquid water.
You can refer to the following chart to see how the volume of water changes with temperature:
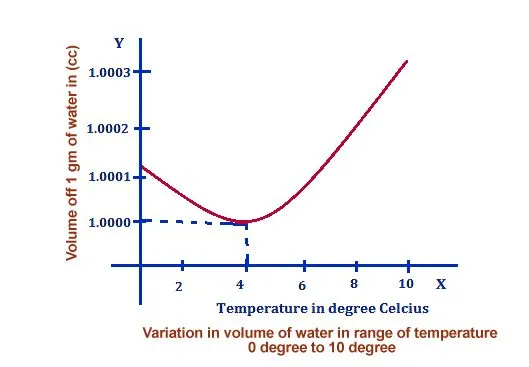
Now, let’s talk about why the volume of water increases or why it expands when it freezes and reaches a solid form.
Why Does The Volume Of Water Increase When It Freezes?
This phenomenon has to do with the chemical constitution of water. You see, a molecule of water is made of two hydrogen atoms and one oxygen atom. The arrangement of these atoms is quite unique, which gives water some special properties, such as the high heat capacity of the water, surface tension, adhesion, and cohesion.
This arrangement of a water molecule creates a positively charged side near the hydrogen atoms and a negatively charged side near the oxygen atom.
When two water molecules come close together, the positive side of one molecule clings to the negative side of the other molecule. When this happens on a large scale (i.e. with millions of water molecules), what you get is a unique structure, which accounts for some of the chemical properties of water.
In liquid form, water molecules can move freely, constantly forming and breaking hydrogen bonds in the process — a property that accounts for the shapeless nature of water (or any liquid, for that matter). Because the bonds keep flickering on and off, the molecules can slide into the gaps between their neighbours and pack quite tightly, which is why liquid water is denser than ice.

However, as the temperature drops and the water cools down, the intermolecular forces increase, the freedom of movement of water molecules decreases, and they become less and less energetic (with decreasing temperature).
When water reaches its freezing point, the movement of its molecules slows dramatically and they lock into a rigid, six-sided (hexagonal) lattice known as ice Ih — the form of ice you encounter in everyday life. In this structure, every water molecule is hydrogen-bonded to four neighbours in a tetrahedral arrangement, leaving large hexagonal channels of empty space running through the crystal.
Below is an oversimplified version of the arrangement of water molecules in their crystalline form in ice:
This crystalline arrangement of water molecules is less dense, as it prevents molecules from huddling up (as happens in the liquid form) due to stronger intermolecular forces.
This spacing of molecules and keeping them fixed in that position increases the volume of water, which is why it’s said that water expands when it freezes.
This Is Why Ice Floats On Water
Water expands when it becomes ice, and since the volume is inversely proportional to the density of a substance, ice is less dense than water. For this reason, ice, a substance that appears heavier than its liquid form, floats on water.
If water didn’t expand when it froze, then ice would be denser than water. Think of the impact on the ecosystem of the planet! Ice on the surface of lakes, seas and oceans would sink and these bodies of water would gradually fill from bottom to top. With frozen lakes and oceans, there would be no aquatic life on Earth.
From that perspective, it’s a very good thing that water expands in its solid form!
Do Other Liquids Expand When They Freeze?
Water is unusual, but it is not entirely alone. Most liquids contract when they freeze because their molecules slow down and pack together more tightly. A handful of substances, however, share water’s quirky habit of expanding upon solidification — typically because, like water, they form open, tetrahedrally-coordinated crystal structures.
The other elements that expand when they freeze include silicon (~2.6%), gallium (~3.1%), bismuth (~3.3%) and antimony (~5.6%). None of them, however, comes close to water’s ~9% jump. So while alcohol, oil and most other household liquids will shrink if you put them in the freezer, water — and a small club of curious metals and metalloids — go the other way.
References (click to expand)
- Water Density, Specific Weight and Thermal Expansion Coefficients — The Engineering ToolBox
- Thermal Expansion and Density | EARTH 111: Water — The Pennsylvania State University
- Water in its three phases — tornado.sfsu.edu
- Ice and Water — Chemistry LibreTexts
- FAQs About Water and Steam — International Association for the Properties of Water and Steam













