Diamond scores a perfect 10 on the Mohs hardness scale, indicating that it is the hardest natural material when subjected to scratching. However, lonsdaleite (hexagonal diamond), a substance found in meteorites, was confirmed in 2025 to be marginally harder than diamond.
Ask any science-loving geek “what is the hardest material?” and he will undoubtedly reply diamond.
The word diamond has its roots in the Greek word ἀδάμας (read as adamas), which means insurmountable or unbreakable. For decades, humans have been harnessing the impeccable hardness of diamond to conduct intensive cutting activities. Also, given its ability to interact beautifully with light, diamonds are a deeply coveted jewelry item for women. But is diamond really the hardest material on Earth?
Well, almost… but scientists have discovered several materials that can rival or even surpass diamond's hardness.
Recommended Video for you:
Naturally Occurring Hardest Substance On Our Planet
When it comes to the naturally occurring hardest substances, diamond is the clear winner. Thanks to its compact structure, it is very hard to beat its hardness. Now, the question arises… how do we measure hardness?
Measuring Hardness
In material science, assessing the hardness of a material is very important. However, hardness isn’t all that easy to define. Thus, hardness can be measured in a different manner, depending on the context and applicability.
Mohs Scale Of Hardness
One of the most commonly used scales of hardness is the Mohs scale of hardness, designed by a German mineralogist Friedrich Mohs in the nineteenth century. In this scale, hardness is a measure of resistance exhibited by a given material when scratched with another. The Mohs scale of hardness ranges from 1 to 10, with 10 indicating the hardest (least affected by scratches) and 1 being the least hard (talc).

Diamond scores a perfect 10 on this scale, clearly indicating that it is the hardest natural material when subjected to scratching. To understand how good a diamond is, consider steel, which is known for its toughness, scoring only a 4.5 on this scale!
Now, measuring hardness by a substance’s scratch-resistant abilities wasn’t approved by everyone. Thus, scientists started looking for an alternate method of measuring hardness. Another hardness technique was developed in which an indenter was used to assess hardness.
Vickers Hardness Test
One of the most famous tests for determining hardness using an indenter is the Vickers hardness test. In this hardness testing method, a pyramid-shaped indenter is pressed against the material whose hardness needs to be evaluated. A specific amount of force is applied for a specific time on the material in question. After this indenting activity, the degree of indentation on the material is measured. This is done by measuring the surface area of the indentation inflicted on the material by the indenter. Here again, diamond was found to be the hardest naturally occurring material on Earth.
What Makes A Diamond So Hard?
At this point, you may be asking yourself, what makes a diamond so hard? The answer lies in the molecular structure of this brilliant gemstone. Diamond is an allotrope of carbon in which each carbon atom is covalently bonded to four neighboring carbon atoms in a tetrahedral lattice structure. These covalent bonds are extremely strong and very tough to break at room temperature.
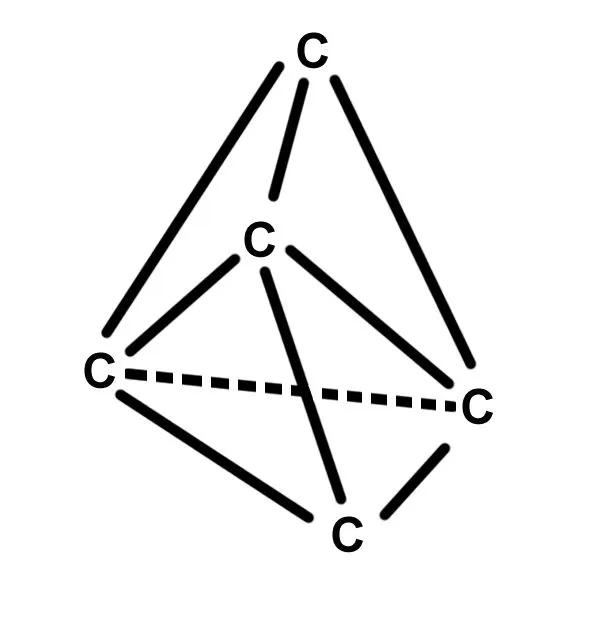
Due to this strong covalent bond, diamonds don’t have free electrons, making them a poor electric conductor, but an excellent heat conductor. In fact, a diamond is around five times better at thermal conduction than copper. Due to their fantastic heat conductivity, diamonds are often present in electrical parts, like heat sinkers.
And The Hardest Material Is…
After reading this, you might feel that diamonds are invincible, but that’s not really the case. Diamond becomes vulnerable at very high temperatures. When you heat a diamond above 800oC, its chemical and physical properties no longer stay the same. A diamond’s characteristic toughness is compromised. They begin to chemically react with iron, which is what makes diamond undesirable for machining steel.
Therefore, scientists and researchers have long been looking for a super-hard material that has better chemical stability. In 2009, researchers working in collaboration at Shanghai Jiao Tong University and the University of Nevada used computer simulations to predict two materials that could beat diamond at its own game: wurtzite boron nitride (w-BN) and lonsdaleite. However, subsequent experimental testing has painted a more nuanced picture.
Wurtzite Boron nitride (w-BN)
Wurtzite boron nitride (w-BN) has a similar structure to that of a diamond, but it’s composed of atoms of boron and nitrogen, alongside carbon. w-BN is extremely rare and can only be found in the aftermath of a certain type of volcanic eruption. A simulation conducted by researchers in 2009 predicted w-BN to be 18% harder than diamond. However, when w-BN was physically tested in 2019 (Liu et al., Scientific Reports), its measured hardness was only about 46 GPa — significantly softer than diamond’s ~110 GPa. The simulation’s prediction did not hold up to experimental verification. That said, w-BN is chemically more stable than diamond at high temperatures.
Lonsdaleite
Lonsdaleite, also known as hexagonal diamond, is made of only carbon atoms, just like a diamond, albeit with a different crystal structure. Lonsdaleite was originally identified in graphite-rich meteorites that struck the Earth. The 2009 simulation predicted lonsdaleite to be 58% harder than diamond, which would have made it the hardest substance on Earth by a wide margin.
For years, scientists debated whether lonsdaleite even existed as a distinct mineral — a 2014 study in Nature Communications argued that supposed lonsdaleite samples were simply faulted cubic diamond. This controversy was finally resolved in 2025, when researchers led by Shoulong Lai at Zhengzhou University successfully synthesized millimeter-sized pure hexagonal diamond crystals by compressing graphite at 20 GPa and 1,300–1,900°C. Their measurements showed lonsdaleite has a Vickers hardness of about 114 GPa — marginally harder (~4%) than natural diamond's ~110 GPa, but far from the 58% predicted by the earlier simulation.
The Reality Check
Now that both materials have been physically tested, the picture is clearer: w-BN turned out to be softer than diamond, while lonsdaleite is only marginally harder — not the dramatic improvement the 2009 simulations predicted. However, lonsdaleite does have a higher Young’s modulus (1,229 GPa vs. diamond’s 1,087 GPa), meaning it is stiffer than diamond.
Interestingly, another material currently holds the title of hardest confirmed substance: aggregated diamond nanorods (ADNR), synthesized from fullerite (compressed C60 molecules) under extreme pressure. ADNR has a measured Vickers hardness of about 150 GPa — harder than single-crystal diamond — and can actually scratch natural diamond. If produced in larger quantities, materials like ADNR and lonsdaleite could be used as powerful cutters by placing them on top of other cutting tools. Their stability at higher temperatures would also make them useful in space missions to Venus or Mercury, which have scorchingly high temperatures.
Closing Thoughts
While diamond has technically lost its crown — with aggregated diamond nanorods and lonsdaleite both measured to be harder — it remains king of the gemstones and is still the hardest naturally occurring mineral on Earth. The story of super-hard materials is a fascinating reminder of how scientific understanding evolves: bold simulation predictions from 2009 were tempered by experimental reality, while entirely new materials like ADNR emerged as unexpected champions. Metaphorically, diamond is under tremendous pressure from its carbon-based peers to maintain its status. Fortunately, diamonds were only able to form because they valiantly withstood extreme external stress beneath tons of rock, so they’re used to handling pressure!
References (click to expand)
- Pan, Z., Sun, H., Zhang, Y., & Chen, C. (2009, February 6). Harder than Diamond: Superior Indentation Strength of Wurtzite BN and Lonsdaleite. Physical Review Letters. American Physical Society (APS).
- Mohs' Scale of Hardness - Geology - rocks and minerals. rocksminerals.flexiblelearning.auckland.ac.nz
- FRONDEL, C., & MARVIN, U. B. (1967, May). Lonsdaleite, a Hexagonal Polymorph of Diamond. Nature. Springer Science and Business Media LLC.