Table of Contents (click to expand)
Diesel and petrol are both obtained from crude oil, but diesel is more efficient and suitable for steady motion because it is denser and ignites more slowly than petrol. Diesel engines are also sturdier and heavier than petrol engines, but they require more maintenance.
Both petrol and diesel fuels are derived from crude oil that is found deep in the earth’s crust. Although both types of fuel are commonly used, diesel is generally used to power larger engines, such as those found in trucks, boats, and cranes, while petrol is used to power smaller engines, such as those found in cars and motorcycles.
Have you ever wondered why this is the case?
Also, what about diesel fuel, which makes it more efficient and suitable for steady motion, while petrol often causes jolting acceleration?
By understanding the differences between these two fuels, we can better understand the various aspects and answers to the abovementioned questions.
Recommended Video for you:
The Difference Between Diesel And Petrol
Crude oil is made up of a mix of different hydrocarbons. These hydrocarbons can form either long, straight chains or rings. The boiling point of a chain or ring is directly related to its size – larger chains require more energy to break apart.
This property can be used to refine or separate different types of hydrocarbons from crude oil.

Fractional Distillation
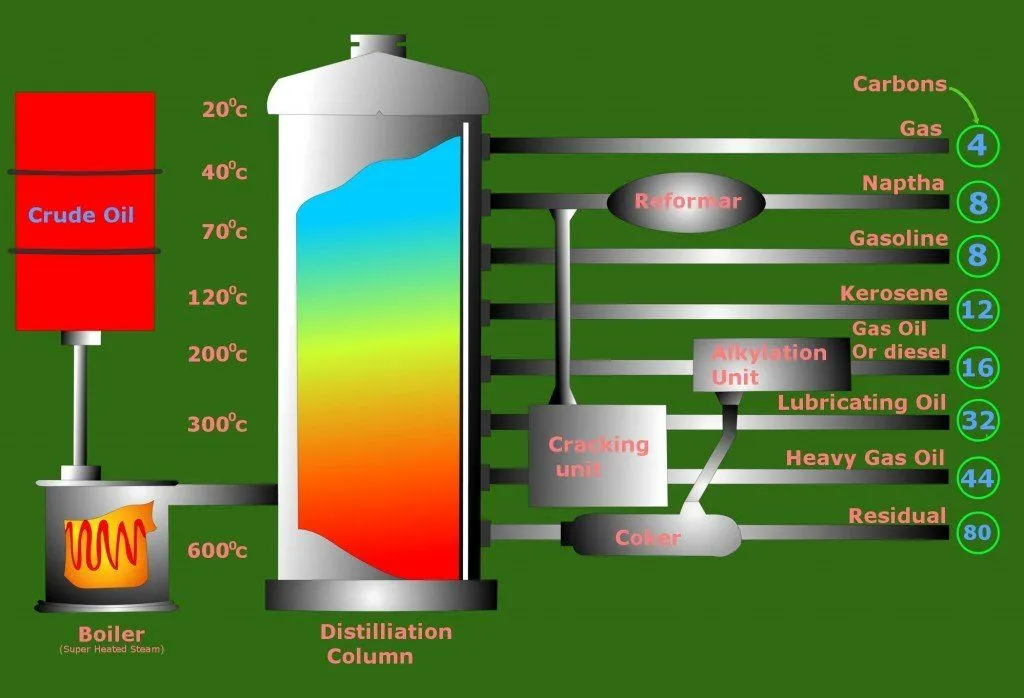
The extracted crude oil is first poured into a gigantic furnace. The furnace or column is then heated. This causes fractional distillation, which separates the crude oil into different components based on their boiling points. The furnace is heated to different temperatures along its length, with the hottest section at the bottom.
The different boiling points of the components allow them to be separated into various fuels. Lighter fuels like LPG and petrol are obtained from the top of the furnace, while denser fuels like diesel are obtained from the middle. The heaviest compounds are obtained from the bottom and can be used as lubricants.
Let me explain the whole process with a salad analogy.
A Salad Analogy
Think of crude oil like a big fruit salad. You want to separate it piece by piece, and you do that using heat in a big furnace, similar to a big oven. This is called fractional distillation, almost like sorting salad pieces by size or color.
The furnace is made to vary in temperature, and it’s like a slide, starting with hot lava at the bottom and ending with a cool breeze at the top. The pieces with different boiling points, like how different fruits can take different cooking times, separate themselves in this furnace slide. Lighter ones that heat up quickly, like LPG and petrol, move to the top, just as small fruit pieces might float in water. Meanwhile, the denser ones that need more heat, like diesel, stay in the middle.
At the bottom, the heaviest pieces stick like heavy fruits sitting in the bottom of a bowl. These heaviest pieces can be used to make things like lubricants, so think of it as using a heavy melon to make a smoothie!
It is now obvious why petrol can cause shoulder-jolting acceleration: as a consequence of being much lighter, it ignites much more quickly than dense diesel fuel. However, due to being denser, diesel packs more hydrocarbons and, therefore, more energy per unit of volume than petrol.
Typically, petrol comprises a recurring chain of no more than nine carbon atoms, while chains constituting diesel contain at least fourteen carbon atoms. Diesel cannot evaporate as rapidly as petrol, but being more glutinous (it is for its viscous, oily texture that it is often called diesel oil), it only packs 132 x 106 J per gallon, while petrol packs 155 x 106 J per gallon. Consequently, vehicles fueled by diesel are more mileage-oriented than vehicles fueled by petrol.
However, while a truck might not generate heart-racing RPMs, it does produce astonishing torque and, therefore, power – the capability to move massive loads. A smaller vehicle’s inability to generate such Herculean torque is attributed to its engine. The operation of a diesel engine is significantly different from that of a petrol engine.

Petrol Vs Diesel: A Detailed Comparison
Petrol ignites more readily than diesel does. Light a match, drop it in a puddle of petrol, and witness how it lights ablaze almost instantaneously. It is for this reason that petrol engines utilize spark plugs. In a petrol engine, the mixture of air and petrol is compressed by a piston and ignited by a spark. Diesel fuel, however, is not so flammable. In a diesel engine, the air is compressed first, and then the diesel is injected. Compressed air combusts, and the air in a diesel engine is compressed so severely that the diesel fuel ignites as soon as it is injected.
Steady ignition demands more compression, which produces more heat and, therefore, a greater capability to do work. The steady ignition has another advantage: it ensures the fuel is leveraged. The amount of chemical energy converted into mechanical energy by a diesel engine is far greater than that of a petrol engine. Consequently, the efficiency of a diesel engine is far greater than the efficiency of a petrol engine.
Let me say this in simpler words.
Petrol catches fire more easily than diesel. It’s like how paper catches fire quicker than a log of wood. If you throw a match into a pool of petrol, it lights up in no time. That’s why cars that run on petrol have spark plugs, which kind of work like a matchstick to ignite the petrol-air mixture in the engine.
On the other hand, diesel isn’t as quick to burst into flames. In a diesel engine, air is squished tight first, and then diesel is added. When you squeeze the air like this, it can cause a fire. The air in a diesel engine is squeezed so hard that it makes the diesel catch fire just after it’s added.
This way of catching fire needs more pressure, but this creates more heat and gives more power to run things. Also, it ensures that all diesel fuel is used.
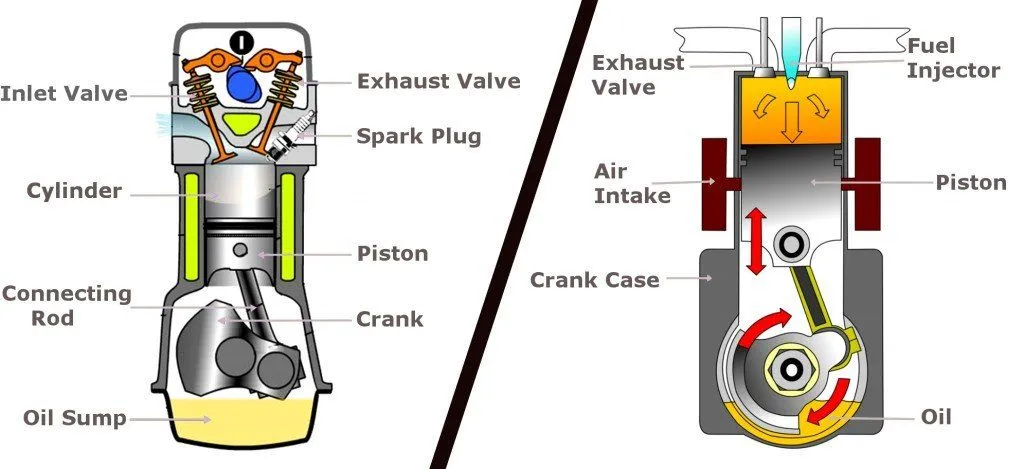
However, an engine must possess great strength to generate and sustain such great torque. Diesel engines are, therefore, relatively sturdier and heavier.
The problem is the heavier the equipment, the greater the intensity of its vibration. The vigorous vibration makes it more liable to wear and tear. As a consequence, diesel engines generally require more maintenance than petrol engines.
Furthermore, diesel engines are constantly stuck in a vicious cycle due to their mass. With great mass comes great resistance to motion, or inertia, so the diesel engine must achieve greater compression to generate a greater torque to move this mass. However, generating and sustaining this greater torque requires even heavier components, which, of course, increase its mass!
What’s more, the combustion of diesel produces dense smoke, or what is called smog and soot – black particles spawned by partial combustion. (Remember that, according to the first law of thermodynamics, an engine can be very efficient but not fully efficient.) Smog is what causes acid rain, while soot is detrimental to our eyes and skin. However, more alarmingly, it can prove fatal if inhaled.
Naturally, shouldn’t we opt for petrol? Well, not really. Petrol is equally detrimental. For producing carbon dioxide and carbon monoxide upon being combusted, petrol suffers universal hatred as the primary cause of global warming.

However, the fuels are indispensable to us despite their notorious reputation. All three sectors of economy and industry — primary, secondary, and tertiary – would become defunct without diesel. It fuels agricultural machines, industrial machines, trucks, cranes, public transport, and every other large-scale machine you can name. Petrol is what fuels cabs, cars, small boats, and every small vehicle you can name.
Keeping this in mind, engineers seek innovations in technologies concerning refining the oil and the engines combusting the procured fuel. After diesel is extracted, it is purged of compounds, the combustion of which produces nitrogen and sulfur – gases that cause acid rain.
This process is rightly called purification. Innovations in constructing petrol engines include units that absorb or, at least, mitigate carbon emissions. Diesel engines, because they vigorously vibrate, are raucous and, therefore, worsen the already woeful noise pollution caused by vehicles.
However, owing to sound and mechanical engineering innovations, engines are quieter and require less maintenance today than they used to.

In fact, burgeoning technological advances — which show no signs of slowing — will eventually render combustible fuel obsolete. A dramatic revolution is incipient. We have just begun to jog; soon, we’ll begin to sprint and leap. The future is not flammable but electric. Posterity will drive (or fly?) cars that are mean and clean. Perhaps these cars will drive themselves.
Last Updated By: Ashish Tiwari












