Table of Contents (click to expand)
The main reason mercury is used in thermometers is because it has a higher coefficient of expansion than water. This means that changes in its volume with temperature are more noticeable. Additionally, mercury has a boiling point of 356.7 degrees Celsius, which is much higher than alcohol’s boiling point of 78.37 degrees Celsius. Finally, mercury is not toxic like alcohol can be.
In principle, any substance whose properties visibly change with temperature can be used to measure temperature. We could have used a material that changes color when subjected to heat. One could discern that the temperature is high when the material would radiate hues of blue and that the temperature is low when it would radiate hues of red.
Similarly, water in a narrow tube rises or falls when its temperature rises or falls. This is the working principle of every sealed, liquid-containing thermometer. However, if something as abundant and cheap as water works so well, why do we insist on snubbing it for something as extremely rare and expensive as mercury?

Recommended Video for you:
History
Mankind learned about thermodynamic temperature centuries after the first thermometer was invented. Therefore, initially, thermometers defined temperatures. The first thermometers, however, weren’t thermos or heat measurers, but thermo-scopes, devices that merely signaled whether the temperature was high or low. These devices weren’t calibrated to a standard scale; they would just make crude or vague assessments.
The invention of the first thermometer is credited to Hero of Alexandria, a curious engineer who is considered the greatest experimenter of antiquity. His device consisted of a tube filled with air whose end was submerged in a tiny bowl of water. When the thermoscope touched a hot or cold surface, the air would expand or contract, causing the air-water interface to fluctuate.

Even Galileo’s invention worked on the same principle. However, not only were these developments bereft of any scale, but they were also sensitive to the air’s pressure. The urge to develop a device that solely responded to heat led Joseph Delmedigo, a student of Galileo’s, to invent the first sealed liquid-in-glass thermometer. This was the first thermometer because it was marked with a scale. However, the liquid he sealed wasn’t water, but alcohol.
Coefficient Of Expansion
Materials, when under constant pressure, expand when subjected to heat because the heat elevates the kinetic energy of its atoms, causing them to violently move and therefore separate from each other. The increase in volume is evident in solids, such as metallic railway tracks, rubber tires and fluids like water, alcohol, mercury and halogen. However, the amount of expansion per degree increase in temperature is different for every material. This material constant is called the coefficient of expansion.

Alcohol is favored over water for the simple reason that it boasts a higher coefficient of expansion. Even a small change in temperature causes a drastic change in its volume along the tube. However, alcohol is so sensitive that these changes cause the alcohol in the tube to behave almost turbulently. The levels constantly waver with even minor changes in temperature. This capriciousness is disconcerting, as the reading would immediately change when, say, the thermometer is removed from a pot of boiling water whose temperature we wished to measure. It would then immediately reflect the temperature of its new environment.
To avoid this unreliability, Dutch inventor Danie Fahrenheit replaced alcohol with mercury. Mercury has a higher coefficient of expansion than water, which means that changes in its volume with temperature are more noticeable. However, its value is almost six times less than that of alcohol. For perspective, the rise or fall in alcohol’s volume per degree rise in temperature would be six times greater than mercury’s volume.
This means that mercury in a sealed tube would rise at a much slower pace than alcohol, but it also means that mercury would fall equally slowly when the thermometer is removed from a pot filled with boiling water. The reading would be effectively undisturbed, making the thermometer superiorly reliable.
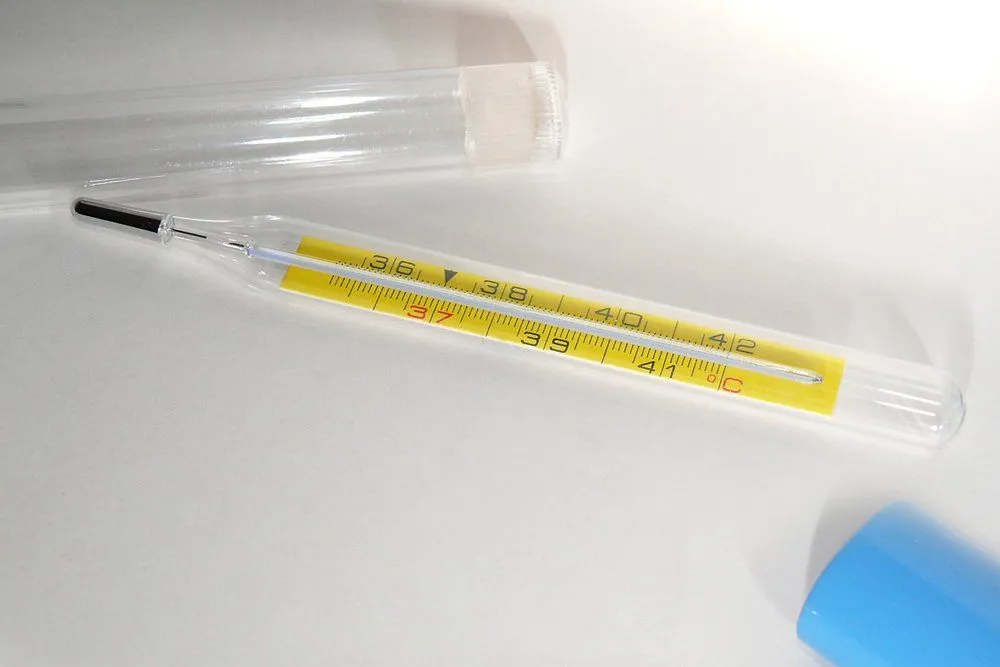
Thermometers before this invention were unique; their readings didn’t correspond to any standardized scale. However, Fahrenheit proposed a scale that was adopted by every manufacturer of mercury and eventually, every type of thermometer. This transition wasn’t exactly cumbersome, since almost every mercury thermometer was manufactured by Fahrenheit himself. The scale, which was slightly altered at a later date, now bears his name.
Instances When Alcohol Fares Better
Alcohol’s hypersensitivity can be compensated by its virtues. Unlike mercury, alcohol is much cheaper and not as ridiculously rare. Also, it is not toxic. A lab might be required to be sealed for hours if a mercury thermometer breaks, considering that inhaling mercury can cause health serious problems. On the other hand, alcohol poses no such threat.
What’s more, alcohol’s freezing point is an astonishing -115ᵒC compared to mercury’s -40ᵒC. This means that mercury thermometers cannot measure temperatures below -40ᵒC, a temperature not all that rare in science laboratories or a superconductor technology manufacturing industry.

However, unlike alcohol, mercury isn’t colorless, a property that forces manufacturers to add artificial dyes to alcohol to make it clearly visible. Also, while alcohol can measure shockingly low temperatures, it is unable to measure temperatures greater than just 78.37ᵒC, alcohol’s boiling point. Compare that meager boiling point to mercury’s incredible 356.7ᵒC boiling point!
While nothing can be done about mercury’s rarity, expensiveness and toxicity, one can still overcome its thermal limitations. To further increase its boiling point, mercury is often sealed with an inert gas, such as nitrogen. The inert gas increases the pressure on the liquid mercury, thereby increasing its boiling point even further.
One can also extend its freezing point by alloying it with thallium. These new mercury-thallium thermometers can now measure temperatures as low as -62ᵒC. Still, despite their small flaws, mercury thermometers are regarded to be one of the most accurate thermometers available.













