Table of Contents (click to expand)
Polaroid pictures use light-sensitive film to capture a scene. The film is developed in seconds, producing a negative image. The negative is exposed to light to create the final positive image. Instant color films work similarly but use three coatings that react to blue, green, and red light and then develop with opposite-colored dyes.
Polaroid cameras were a revolutionary invention by Harvard inventor Edwin Land. Before their invention, developing colored images was a tedious process that took days. Land compressed the darkroom into a single camera and founded the Polaroid Corporation. He made a fortune of $5 million in camera sales in 1949!
Instant cameras are still commonly referred to as Polaroid cameras, and instant film as Polaroid pictures. But how do they actually work?

Recommended Video for you:
How Photographs Were Developed Using Films
At the beginning of the 21st century, we started converting images into pixels and storing them on memory cards. However, in the 1950s, there was no such technology available. Films were used to capture photographs, but they took time to develop.
A film is a plastic sheet coated with silver halides — silver compounds containing halogens (chlorine, bromine, iodine), the elements of Group 17 of the periodic table. These compounds are extremely sensitive to light, so the film is manufactured and loaded into the camera in dark environments to avoid premature exposure.
When you press the button on the camera to capture an image, the shutter opens, and the light-sensitive film is exposed to the scene you want to capture. The light energy disturbs the silver halides covering the film, ionizing the compound and extracting individual metallic silver atoms.

The amount of silver atoms produced is proportional to the amount of exposed light. Therefore, if you capture a tree standing in broad daylight, the area of the film exposed to the sky would have more silver atoms than the area exposed to the tree.
The film’s parts exposed to more light are much darker than the less exposed portions, resulting in a negative image. This negative image is the opposite of the original scene. To create the original or positive image, we must take a negative image of this negative image.
If you want a black-and-white image, you must reel the negative to light. The previously dark parts prevent light from passing through, while the silver halide in the brighter parts of the film ionizes to produce silver atoms, resulting in darker colors. The darker areas are now white, and the brighter areas are now dark. The colors are reversed once again, creating the original, positive image.

Instant Color Films
The same principle is used to develop colored pictures. While the process used to be carried out in labs, Polaroid founder Edwin Land — who introduced the first commercial instant camera, the Model 95, in 1948 — compressed the lab into the camera itself. Instant film is covered with not one but three silver halide coatings. The first coating (from up to down) responds to blue light, the second to green light, and the third to red light.
However, remember that the films produced are negatives when blue, green, and red lights fall onto these coatings. Silver atoms in the plates are densely accumulated in areas exposed to more light. In terms of color, the third plate encoded with the information of red light is actually cyan.
Similarly, the other two plates are also painted in ‘opposite’ colors: the green plate is covered in magenta, and the blue plate is covered in yellow.
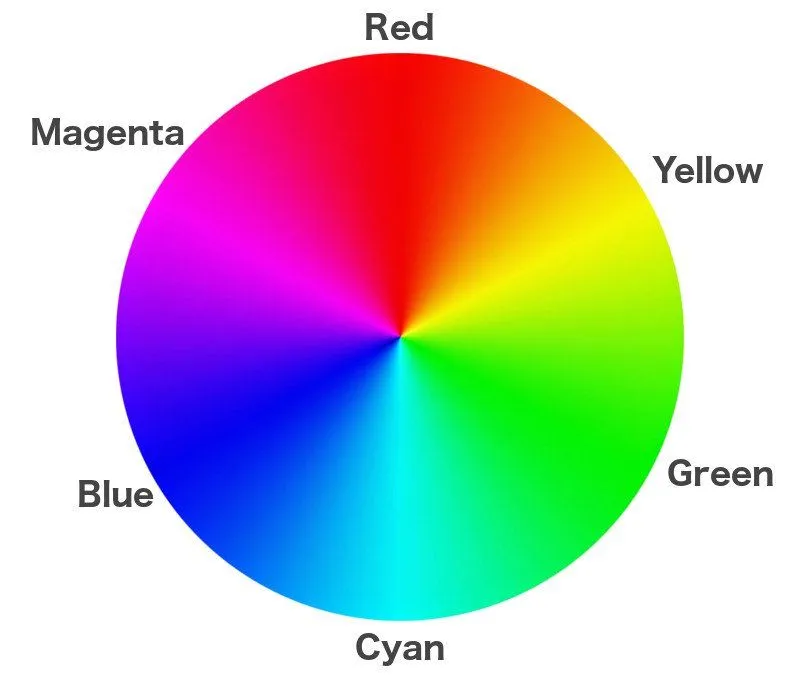
Deposited below each plate is a coating of the opposite color: yellow below the blue plate, magenta below the green plate, and cyan below the red plate. These are known as developing dyes. The last coating at the bottom is black, which ensures that every speck of light is absorbed or none is reflected, as this light could distort the image.
Just like the case of a black-and-white image, the application of cyan to the red implements a negative of the negative. It cancels the red areas and reacts with the unexposed silver halides in the cyan areas to form red. Similarly, magenta and yellow react with the green and blue coatings to implement a negative of the negative.
We know that combinations of red, green, and blue can create any color there is, so the three positive plates are then overlaid to form the original colored image.
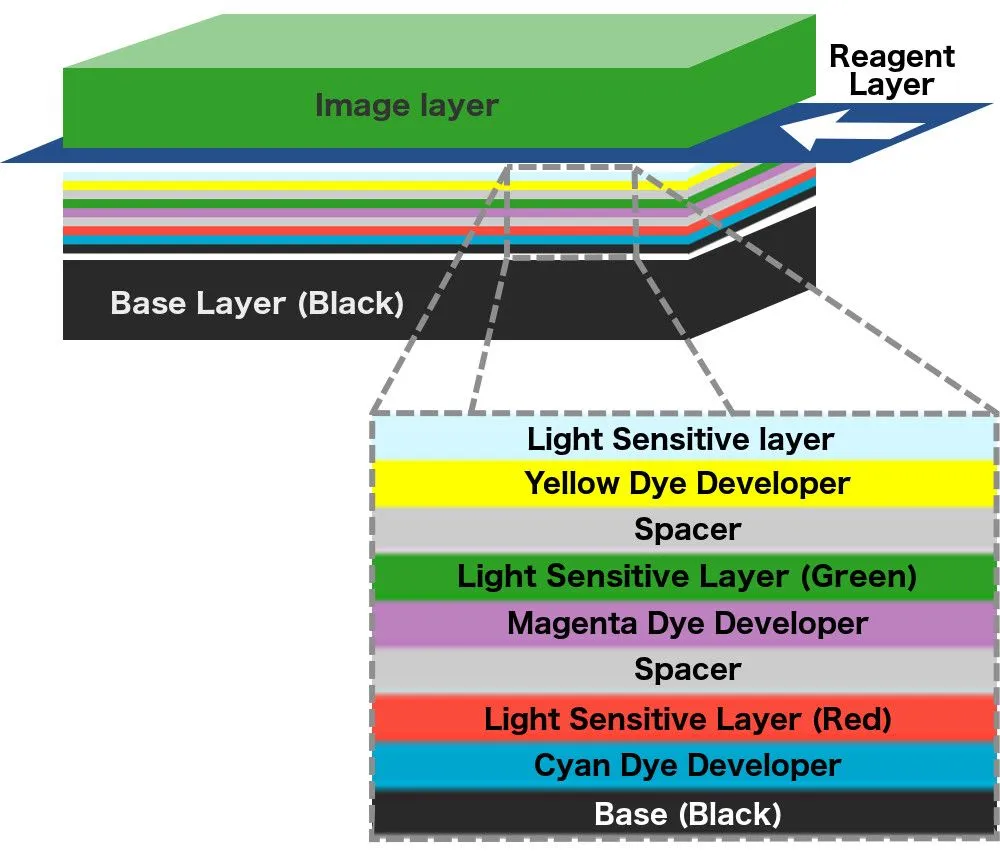
The series of layers stack up like dominoes, and just like a set of dominoes, the chain reaction cannot begin without a trigger. A reactive agent or reagent triggers the chemical reaction that forces the dyes to move upwards. The reagent is stored in pouches that are adhered to the iconic white space below a Polaroid picture. To apply the reagent over the coatings on the film above, a mechanism inside the camera sends the film between two rollers such that the rollers pop the pouch and smear its contents over the film as the film moves forward. In subsequent models, the reagent was applied to another film that was then deposited over the photographic film.
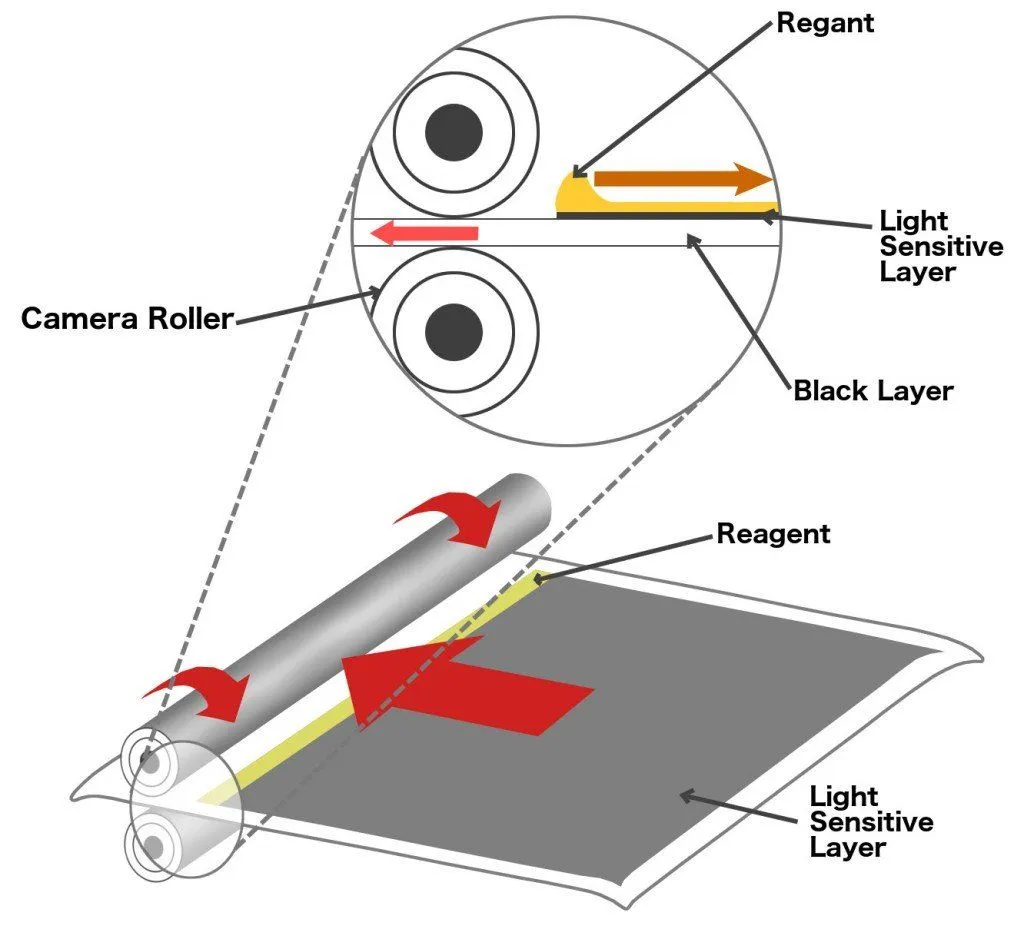
So, as soon as the shutter button was clicked, the film with reoriented silver atoms would be coated with the reagent, and a chemical reaction would ensue. The dyes would flow upwards and color the three coatings.
Finally, almost immediately, as if by magic, the camera would gradually print a piece of white plastic until it would hang out by its end like the camera’s tongue. The dyes need some time to completely dry, so one can smudge the picture with a Q-tip to impart a painting effect. The white plastic’s colors result from incident light reflected through the dyes smeared on its surface.
For the simplicity of Land’s revolutionary innovation, he’s considered nothing less than a genius. Also, contrary to popular belief, shaking the picture does nothing useful and can actually damage modern integral film by separating the wet dye layers — Polaroid itself officially advises against it.
Last Updated By: Ashish Tiwari












