The melting of the polar ice caps is a big threat to the world because it could cause the ocean’s salinity to decrease. This would have a negative impact on the ocean’s currents, which are important for bringing food and nutrients to marine life.
You might already know that one of the biggest threats associated with climate change and an increase in the average temperature of the globe is the melting of the polar ice caps. As the average temperature increases worldwide and the Earth becomes increasingly hotter, it’s obvious that the gigantic ice caps would start melting too (as they already are), and the water from all that melting would join the waters of the world’s oceans.

What happens when you add water to a saline aqueous solution? It becomes less salty, right?
You already know that seawater is salty, so what do you think would happen when it suddenly gets a huge supply of freshwater (i.e., non-salty water)? Would it become less salty?
As it turns out, the answer is not that simple, and neither are the consequences of such a situation. First, let’s cover a few basics.
Recommended Video for you:
What Is Salinity?
Salinity, in simple terms, refers to the dissolved salt content of a water body. As you can imagine, seawater’s salinity plays a crucial role in determining the type organisms that can thrive in it. The salinity of seawater plays an instrumental role in ocean circulation and the water cycle.

The salinity of seawater is dependent on a few factors, including evaporation, rainfall, wind, the inflow of river water and the melting of glaciers. For the scope of this article, we’re only going to discuss the last factor.
The Impact Of Global Warming
Quite predictably, the heating of the Earth’s surface is resulting in the melting of polar ice caps in Antarctica and other polar regions. Research also shows that the Arctic Sea ice is melting faster than expected. This is because as the Earth continues to heat as a result of global warming and the Arctic ice melts, the influx of freshwater from the melting ice changes the salinity of seawater, especially at high latitudes.
Therefore, it’s no surprise that the salinity of all the world’s oceans is decreased as a result of melting polar ice caps. The Atlantic Ocean, in particular, has seen quite a significant change in its water’s salinity over the past four decades.
So, to answer the question posed in the title of the article, yes, melting polar ice caps make water less salty, but that’s not the complete answer. You see, an increased influx of freshwater into seawater does make the latter less salty, but not in the way you might think.
How Deglaciation Impacts The Salinity Of World’s Oceans
Note that oceans are huge (massive) bodies of water. When such huge water bodies are under consideration, one also needs to understand that the water that these water bodies contain is of different ‘types’. In other words, it takes gargantuan amounts of energy to mix water masses with different properties.
The direct upshot of this is that, instead of the entire ocean reducing its saltiness, certain ocean currents take the biggest hits (i.e., their salinity is the most affected).
Ocean Currents
An ocean current is a directed, continuous movement of seawater that’s brought about by certain forces acting on it. Ocean water is always moving, and ocean currents are movements of water that occur on both the oceans’ surface and their depths, both locally and globally.
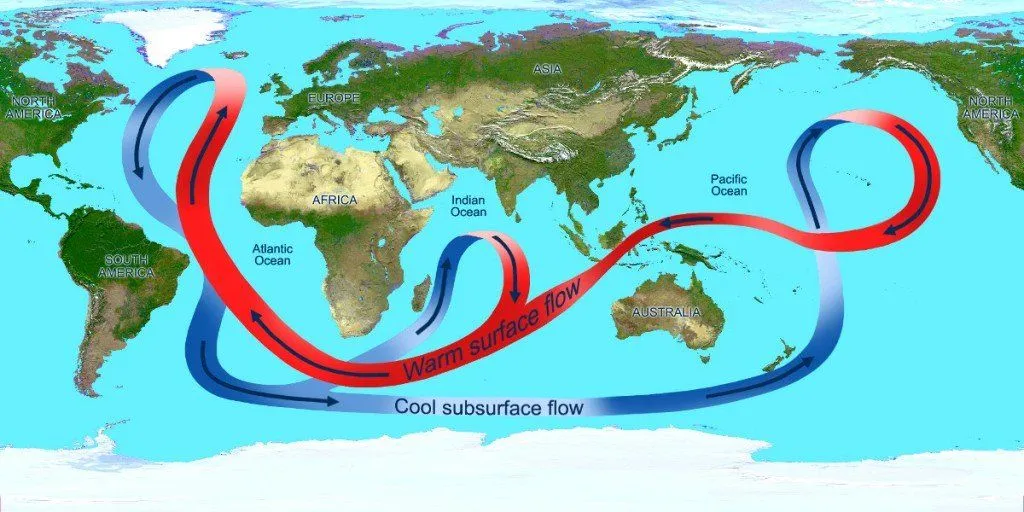
Ocean currents are directed by factors like water density, winds, tides etc. Ocean currents are very, very important, especially to marine life, because many aquatic organisms (with limited mobility) rely on this “liquid wind” to bring food and important nutrients to them. Not only this, but ocean currents also distribute larvae and reproductive cells among marine creatures.
As such, deglaciation, or the mass melting of ice caps, partially stops the exchange of salt, energy and heat between the deep and upper ocean, which leaves only tidally-aided and topography-driven sources of turbulence to help these two layers of the ocean’s water to ‘communicate’.
The Negative Impact Of The Decreasing Salinity Of Ocean Currents
A huge influx of freshwater (as a result of melting ice caps) could alter or even potentially shut down some major ocean currents.

As discussed earlier, these ocean currents are really the ‘good guys’, as they not only distribute food and reproductive cells to distant fish, but also keep the oceans oxygenated so that marine animals can survive in them.
Any negative impact on these ocean currents could seriously disrupt the food chain in oceans, which would undoubtedly have ramifications on the food chain that we humans also exist within.
So, yes, the melting of ice caps surely reduces the salinity of oceans (albeit not uniformly), and this melting is much more dangerous to humans than many people expect.
References (click to expand)
- How Melting Arctic Ice Affects Ocean Currents. The University Corporation for Atmospheric Research
- Roberts, N. L., Piotrowski, A. M., McManus, J. F., & Keigwin, L. D. (2010, January). Synchronous Deglacial Overturning and Water Mass Source Changes. Science. American Association for the Advancement of Science (AAAS).
- Flament, P. (2002, July). A state variable for characterizing water masses and their diffusive stability: spiciness. Progress in Oceanography. Elsevier BV.
- Ocean Stratification - eesc.columbia.edu
- Ocean currents - NOAA. The National Oceanic and Atmospheric Administration