Table of Contents (click to expand)
LPG gas is basically propane and butane, and it is odorless in its natural state. The smell that you notice when there is a leak actually comes from an entirely different agent, called Ethyl Mercaptan. This substance is added to the gas when it leaves the main storage terminals.
You are likely familiar with the stench that turns up your nose if there is a leak from an LPG (Liquid Petroleum Gas) cylinder nearby. It’s quite interesting to note, however, that LPG itself is actually odorless!
Recommended Video for you:
What Is LPG?
Liquefied Petroleum Gas contains propane, propylene, butane, and butylene in a number of mixtures and concentrations. It is completely odorless, and is made from refining petroleum or natural gas. The source is typically fossil fuels and related sources, although they can also be extracted from petroleum or natural gas streams. The gas is kept in a liquid state by using high pressure and is transported via pressurized steel vessels.
The most common uses of LPG include rural heating, cooking, motor fuel and refrigeration. Environmentally LPG is a better alternative too! It burns more cleanly than heavier molecular weight substances, as it releases less particulate matter. It does, however, release carbon dioxide and a small amount of carbon monoxide. The amount of carbon dioxide per kWh released by LPG is around 81% that of oil and 70% that of coal!
LPG is one of the most convenient fuel sources for cooking and also doesn’t burn a hole in the user’s pocket. North America, India, and some Brazilian urban areas are major users of LPG. For heating, LPG has primarily been used in Europe as an alternative to electric heating and heating oil.
Also Read: Can CNG And LPG Be Used As Car Fuels?
What Gives LPG Its Characteristic Odor?
Ethyl Mercaptan
LPG gas is basically propane and butane, and it is odorless in its natural state. The smell that you notice when there is a leak is actually from an entirely different agent called Ethyl Mercaptan. This substance is added to the gas when it leaves the main storage terminals.
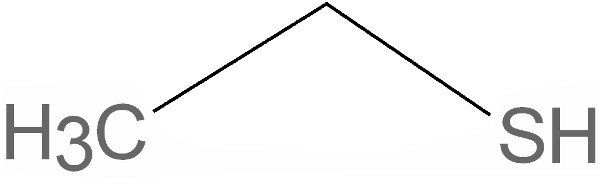
Ethyl Mercaptan, a.k.a ethanethiol, is a clear colorless liquid. It has an overpowering stench with vapors that can irritate the nose and mouth. It can even be lethal if swallowed or inhaled. However, the amount of this substance added to LPG is safe and does not cause any issues.
As we all know, LPG is very dangerous, because if it leaks, it can cause terrible fires and devastating explosions. To avoid this, Ethyl Mercaptan is added to the gas, which possesses that strong odor of rotten cabbages. This smell helps us detect whenever there is a leak, which actually makes a lot of sense from a safety and security perspective. In fact, some people actually refer to the process of adding this foul-smelling substance as “stenching”.
Also Read: Why Do We Like The Smell Of Gasoline?
Pros And Cons Of LPG
The biggest advantage of LPG is the convenience and portability it offers. In fact, this is the primary reason why gas is liquidized into containers in the first place.
In places where ordinary gas supplies cannot reach via gas pipelines, LPG cylinders and tanks offer a convenient and relatively safe alternative to transport gas. This is especially true for hinterlands and rural places, where ordinary gas pipelines simply cannot reach. In addition to that, LPG cylinders prove to be extremely useful during emergencies and natural calamities.
On the other hand, there is certainly a safety concern with LPG cylinders. If you pressurize a gas so much that it occupies 274 times less space than it normally would, you need really sturdy and strong metallic tanks and cylinders that can safely contain such a highly pressurized gas. Despite that, there are the occasional instances of explosions of LPG cylinders and tanks.
Remember that LPG is highly flammable, which means that it can catch on fire quite easily. Logically, one could say that transporting LPG by vehicle through populated areas is more dangerous than piping natural gas underground.
That being said, LPG suppliers ensure the safety of storage and transportation of LPG, making it a versatile and reliable source of energy.
References (click to expand)
- https://www.durhamtech.edu/sites/default/files/safety_data_sheets/Machine-Shop/Propane%20gas%20cas74-98-6%20SDS.pdf
- Campbell, I. D. (1987, August). Odorant Depletion in Portable Cylinders. In Institute of Gas Technology Odorization Symposium.
- ETHYL MERCAPTAN - CAMEO Chemicals - NOAA. The National Oceanic and Atmospheric Admin












