Oxygen is supplied onboard a submarine through pressurized tanks or an oxygen generator, while drinking water is obtained by desalinating seawater.
A submarine is a special ship designed to operate independently underwater for days. The most notable thing about a submarine is that it stays underwater for days. This leads to a natural question – if a submarine is constantly surrounded by water, how does it have enough drinking water for its crew members?

Even more importantly, how do the crew members get breathable oxygen, the most crucial requirement for human life?
Recommended Video for you:
Life Support Systems In Submarines
Due to their multifaceted usefulness, submarines have become essential to naval armies from various countries worldwide. One of the most common applications of submarines is to surveil and defend a country’s maritime borders. Human-crewed submarines are deployed underwater along a nation’s coastal borders to look for suspicious activities in the waters.

This necessitates deploying a considerable number of “manned” submarines underwater. A submarine can typically support around 50-100 passengers. Therefore, you need a system in your submarine that can take care of the water needs of all passengers, maintain comfortable temperatures onboard, and, most importantly, provide fresh, breathable air without interruption.
How Does A Submarine Crew Get Drinkable Water Onboard?
Submarines are surrounded by water almost always, but that’s seawater, which is salty. As such, it cannot be directly used by the crew or even to maintain the engines and batteries of the submarines, which consume around 4000 gallons of water per week. (Source)
To get freshwater, submarines have a distillation apparatus that heats seawater to create water vapor, removing salt. The water vapor is then condensed to create freshwater that can be used for drinking, personal hygiene, and cooking.
How Does The Crew Get Breathable Air Inside A Submarine?
There are different ways to release oxygen on a submarine, such as compressed tanks, an oxygen generator, or an oxygen canister that works through electrolysis. Oxygen is either released periodically throughout the day at specific intervals or when the automated system detects a drop in oxygen levels.
Removal Of Carbon Dioxide
Breathing is a two-way process – we inhale oxygen and exhale carbon dioxide. We don’t need to worry about the carbon dioxide we exhale in open spaces. Still, in a closed metal tube hundreds of feet beneath the water’s surface, it can become a serious threat to survival if not removed periodically. As a result, it’s just as important to eliminate the carbon dioxide that the crew members constantly release as it is to provide oxygen.
This is where scrubbers come in.
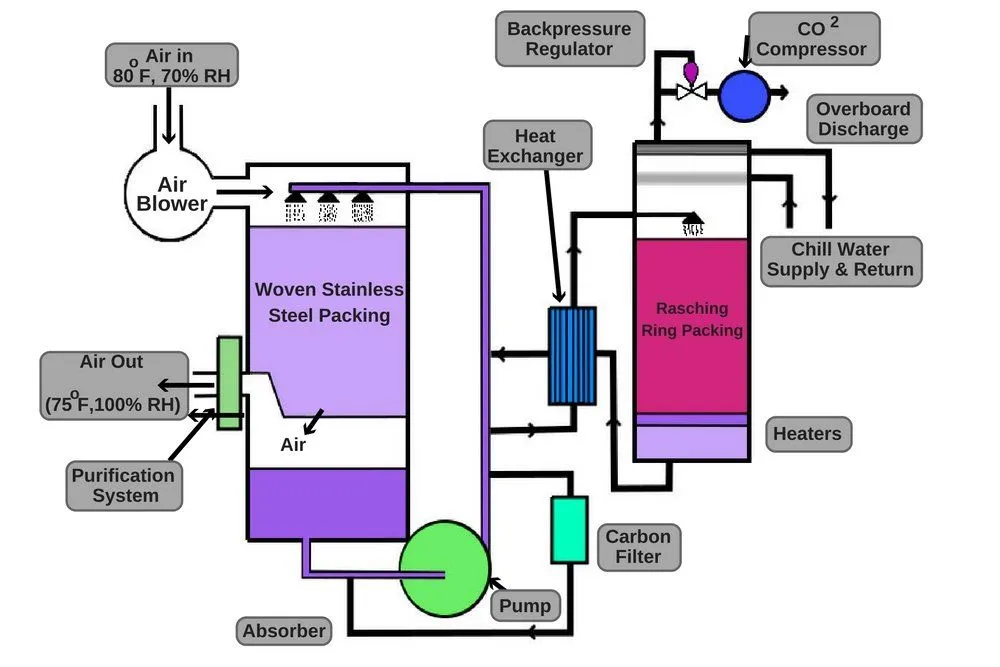
They use soda lime, a mixture of chemicals (sodium hydroxide and calcium hydroxide), to eliminate carbon dioxide from the atmosphere and prevent carbon dioxide poisoning. Soda lime is commonly used in closed-breathing environments, such as submarines, rebreathers, and recompression chambers. It captures the carbon dioxide produced by the crew members and removes it from the air using chemical reactions.
Removal Of Excess Humidity
Excess humidity can harm the inside of a submarine, as it can condense on the walls and equipment. To address this issue, submarines are equipped with ‘dehumidifiers’ that prevent moisture accumulation within the chamber due to breathing and perspiration.
Overall, the safety and comfort of the passengers aboard a submarine are ensured through sophisticated machinery onboard. However, being confined to a metal tube hundreds of feet underwater without sunshine and suffering from homesickness can be a lot less thrilling than it may seem.
Last Updated By: Ashish Tiwari
References (click to expand)
- GELLER, R. E. (1968, November). Oxygen safety - Submarine to aircraft. Journal of Aircraft. American Institute of Aeronautics and Astronautics (AIAA).
- The Machinery of Modern Submarines.
- Lee, D., Lee, J., & Lee, K. H. (2002). A Decision-Support System to Improve Damage Survivability of Submarine. Developments in Applied Artificial Intelligence. Springer Berlin Heidelberg.
- Loveman, G. A. M., & Edney, J. J. E. (2021, February 3). Predicting the effect of decompression sickness on survival following submarine tower escape. Journal of Defense Analytics and Logistics. Emerald.
- Shadle, T., & Daley, T. (1991, July 1). U.S. Navy Submarine Life Support Systems. SAE Technical Paper Series. SAE International.













