Table of Contents (click to expand)
Blackbody radiation has been studied by physicists for years and led to the formulation of quantum mechanics. Now, it has other uses as well.
In our daily life, there is electromagnetic radiation of different wavelengths everywhere around us. One type of radiation our eyes can detect is the visible light by which we see our surroundings. Another kind is the radio waves we use in our communication systems, radios, etc.
Humans have studied and learnt to use electromagnetic radiation in various ways. In that process, we have also mastered ways to produce these radiation emanations.
For example, we produce X-rays (another type of EM radiation) using X-ray tubes. These tubes accelerate electrons and make them hit a stationary target, which in turn causes them to emit X-rays. We use incandescent bulbs and LEDs that emit visible light radiations for us to see objects.
With early discoveries stimulating new avenues of thought, physicists in the 19th century, including those like Gustav Kirchhoff and Ludwig Boltzmann, had a strange idea. They wondered if it would be possible for an object to exist that could emit (or absorb) radiation of all wavelengths. Strangely enough, scientists bought that idea. Several physicists, like John William Draper and John Tyndall, during the early to mid-19th century, conducted experiments on finding out more about the radiation emitted by bodies at various temperatures.
In the late 1850s, Balfour Stewart experimented with a material called lamp-black and said that it had the highest absorbing and radiating strength. It might have served as a precursor to the concept of a black body.
However, Gustav Kirchhoff was the first to define the notion of a black body, in 1859-60. In the subsequent decades, physicists like Wilhelm Wien, Lord Rayleigh and James Jeans made attempts to explain it mathematically. However, they could only partially explain it.

Recommended Video for you:
About A Black Body And Its Radiation
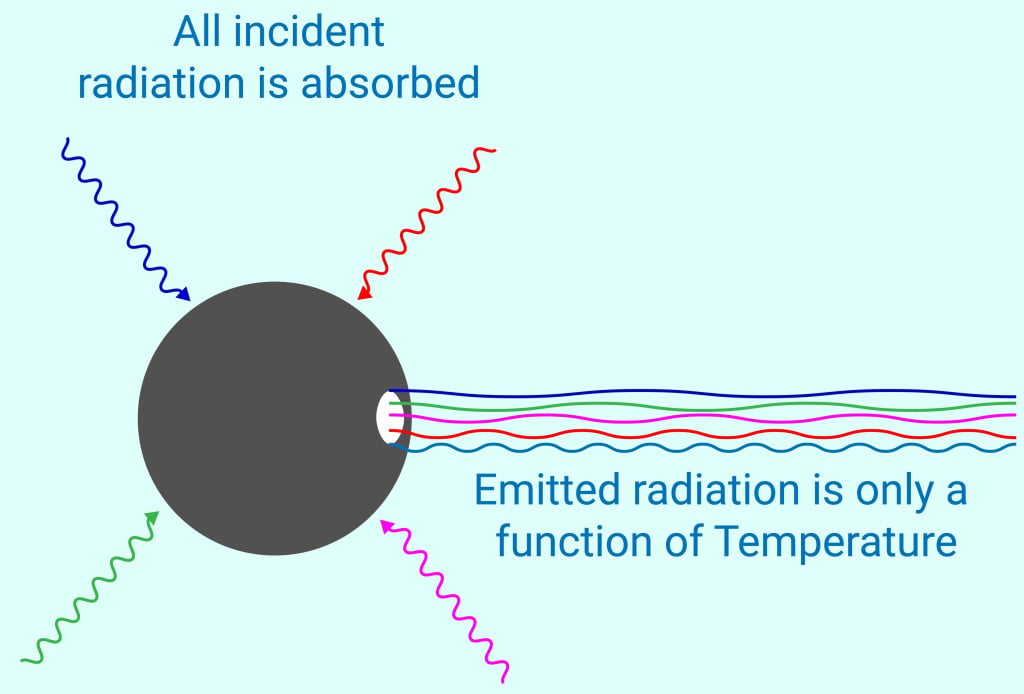
A few things need to be mentioned about black bodies. The scientific world defines a black body as an object that can absorb all radiation falling upon it. It could also mean materials that emit radiations of all wavelengths. The radiations emitted by a black body are called blackbody radiations. The nature of blackbody radiation (like how bright it would appear) would depend only on the temperature of the black body.
Thirdly, and most importantly, an ideal black body is only a hypothetical case, since no object can absorb one hundred percent of radiation falling on it. A real-life body would reflect or transmit at least a tiny amount of radiation through it. However, we have examples of real-world objects that show behavior close to ideal black bodies.
In simpler terms, a “black body” doesn’t always look black. Its color (or the wavelength of light it emits the most) depends on its temperature. Imagine it like a piece of metal that you heat up. At first, the metal glows red/orange, and as it gets even hotter, it turns white and then blue-white. This change in color happens because its temperature affects the wavelength (or type) of light that it primarily emits.
When the black body is extremely hot, the wavelength it emits could even shift to invisible ultraviolet light. On the other hand, if it’s really cold, the wavelength it emits might shift towards infrared or microwaves. In this case, the black body does appear black, as our eyes can’t see those types of light.
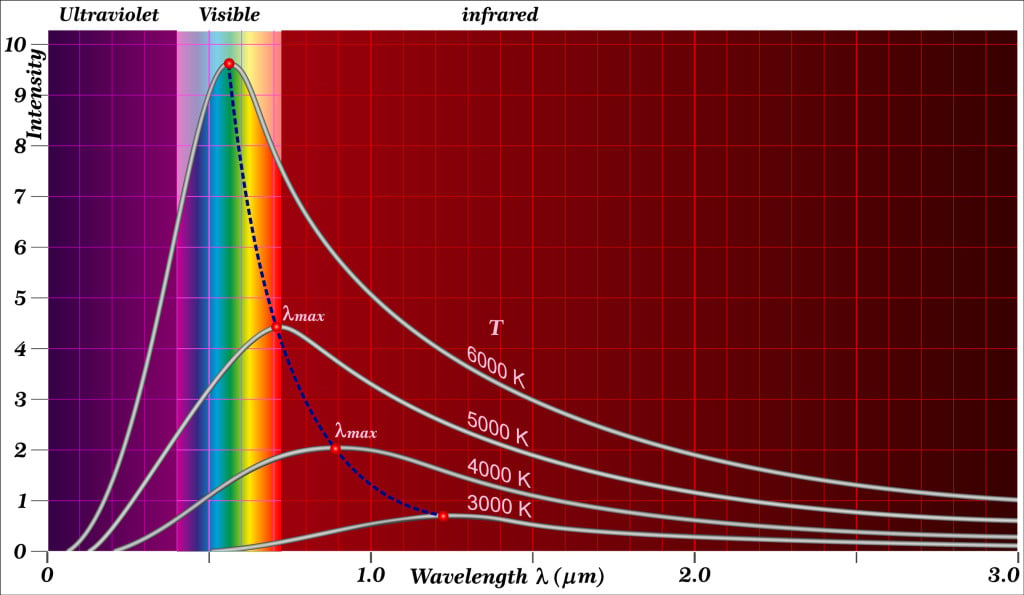
This variation of peak intensity with temperature can be displayed using what physicists call the spectral distribution of blackbody radiation. It is simply a graph of the brightness (or flux) versus wavelengths of a blackbody spectrum at a particular temperature. In other terms, physicists also call this the blackbody spectrum.
By making several such graphs at various temperatures, we see this shift in the peak intensity variation with increasing temperature. Scientists get this plot by measuring the spectrum of objects that show similar blackbody behavior. There is an equation that relates the peak intensity wavelength with absolute temperature. This equation is known as Wien’s displacement law.
Additionally, we can see in these plots that the intensity is never zero for any wavelength. It is related to the blackbody property that it can emit radiations of all wavelengths.
Explaining The Blackbody Spectrum
Many physicists during the late 19th and early 20th century were attempting to provide a theoretical explanation for this plot. From the physics that was prevalent back then (also called classical physics), there was the Rayleigh-Jeans law. This law could, however, only explain this blackbody spectrum for higher wavelengths.
At lower wavelengths, the Rayleigh-Jeans law implies that the intensity would keep increasing sharply, eventually reaching infinity. We do not observe infinite brightness in nature.
Another law that tried to explain the black body spectrum is Wein’s approximation or Wein’s distribution law. In this case, the law could only explain the lower wavelength region of the spectrum. At higher wavelengths, this law did not agree with the observed blackbody spectrum.
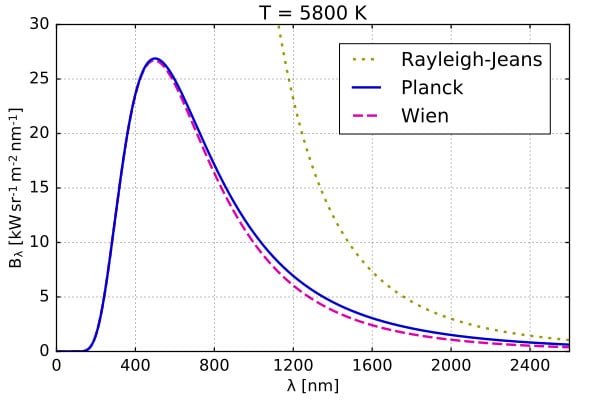
It was not until 1900 when Max Planck, a Nobel Prize-winning physicist, successfully explained the blackbody spectrum for all wavelengths using his quantum theory and radiation law. Its ability to explain the blackbody spectrum became one of the reasons for the success and further development of quantum mechanics.
Black Bodies In Real Life
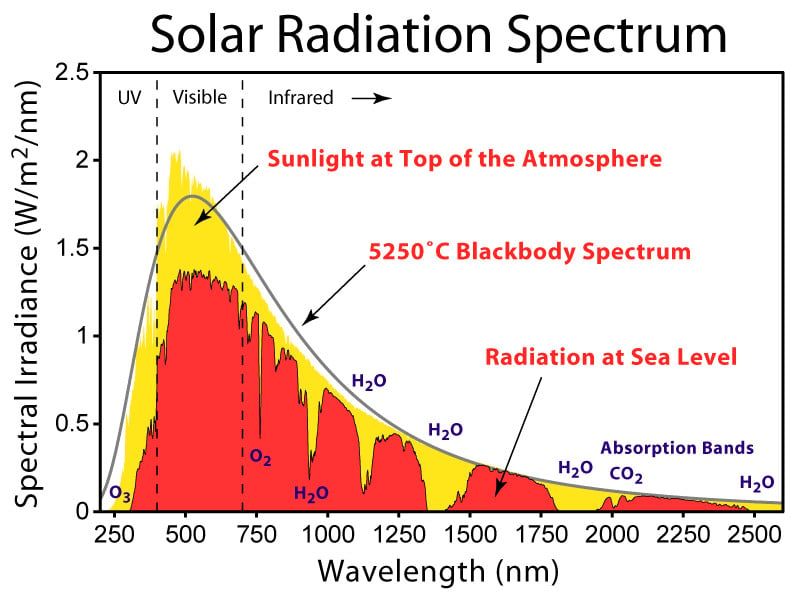
Although no object is one hundred percent black body (that is, it can absorb all the radiation falling on it without reflecting any of it), some objects in nature closely exhibit the blackbody spectrum. For example, the electromagnetic spectrum obtained from stars is similar to that of a black body. In fact, by finding the peak intensity wavelength from that spectrum, the star’s temperature can be obtained using Wein’s displacement law.
Another such example of one found in nature is cosmic microwave background radiation. First discovered by physicist Arnos Penzias and radio astronomer Robert Wilson, the cosmic microwave background radiation closely resembles a blackbody spectrum. Its temperature is about 2.725 Kelvin, giving a peak intensity wavelength of 1 mm.
Apart from this, humans have also attempted to make materials that show properties similar to that of black bodies. One such material developed made use of carbon nanotubes aligned vertically. This material displayed a very high extent of absorption of electromagnetic radiation, similar to what an ideal black body would do.

Scientists obtained similar results using carbon tubes arranged in a dandelion-like arrangement. In this case, the material absorbed more than 99.9 percent of radiation falling on it at various angles.
Another attempt to make a similar material involves chemical etching of nickel-phosphorus alloy. It resulted in a black surface that reflected less than 0.4 percent of the visible light.
The theoretical development of the black body and its radiation drove much of physics at the start of the 20th century. It helped build the foundation of quantum mechanics and helped us discover new theories and laws. Materials developed to behave like black bodies could be used in defense systems to escape radar detection, and the technology may also be used in telescopes to prevent surfaces from reflecting light.
References (click to expand)
- Mavani, H., & Singh, N. (2022). A Concise History of the Black-body Radiation Problem (Version 1). arXiv.
- An open-access textbook for first-year chemistry courses.
- Blackbody radiation in classical physics: A historical ....
- Blackbody Radiation | COSMOS.
- Lecture 38: The Cosmic Microwave Background.
- Blackbody Radiation.
- Production and detection of X-rays.
- Brown, R. J. C., Brewer, P. J., & Milton, M. J. T. (2002). The physical and chemical properties of electroless nickel–phosphorus alloys and low reflectance nickel–phosphorus black surfaces. J. Mater. Chem.. Royal Society of Chemistry (RSC).
- Ghai, V., Singh, H., & Agnihotri, P. K. (2019, November 22). Dandelion-Like Carbon Nanotubes for Near-Perfect Black Surfaces. ACS Applied Nano Materials. American Chemical Society (ACS).
- Mizuno, K., Ishii, J., Kishida, H., Hayamizu, Y., Yasuda, S., Futaba, D. N., … Hata, K. (2009, April 14). A black body absorber from vertically aligned single-walled carbon nanotubes. Proceedings of the National Academy of Sciences. Proceedings of the National Academy of Sciences.
- All material objects emit electromagnetic radiation.