Plants have different shades of green because of different amounts of chlorophyll in their leaves as well as different combinations of other pigments.
Take an adventure out in nature and you’ll find an artist’s palette laid at your fingertips. The gorgeous hues of pink and purple flowers, the firm and reliable colors of browns in the tree bark, and the canopy of green in the leaves rustling in the wind. Within the world of greens, there are so many varieties, fern green, myrtle green, pine green, not to mention mint green, lime green, and avocado green. Teal, olive and dark moss green are just a few more.
Within this wondrous diversity of greens lies a fantastic molecular secret that sustains all life on our planet.
Recommended Video for you:
Plants Have Many Colored Pigments
In biology, color comes from complex organic pigment macromolecules. The green that colors a tree’s leaves is the result of chlorophyll. That word finds its roots in the Greek words chloros (“green”) and phyllon (“leaf”). Sitting within the cells of leaves is an organelle called the chloroplast, with the job of harvesting the sun’s energy. The energy it collects is used by plants to make its food through a process with which we’re all familiar: photosynthesis. It gets its green color from the kind of light it traps energy from.
All pigments get their color by reflecting and absorbing certain wavelengths in natural light. Chlorophyll appears green, since it strongly absorbs wavelengths from the blue and red regions of the electromagnetic spectrum, while reflecting wavelengths from the green, yellow and orange spectrum in varying degrees.
Chlorophyll’s green color can be explained through its molecular structure. Chlorophyll is made of a planar porphyrin ring with a magnesium bonded (coordinately) to the four nitrogen atoms in the porphyrin ring. Attached to the porphyrin ring are different organic moieties. This structure is the reason for its strong absorbance of light in the visible region. The porphyrin ring, along with the coordinate bond with metal ions, makes porphyrin offer a vast range of colors, from the red of heme in hemoglobin (with iron at its core, rather than magnesium) to the greens we seen in chlorophyll.
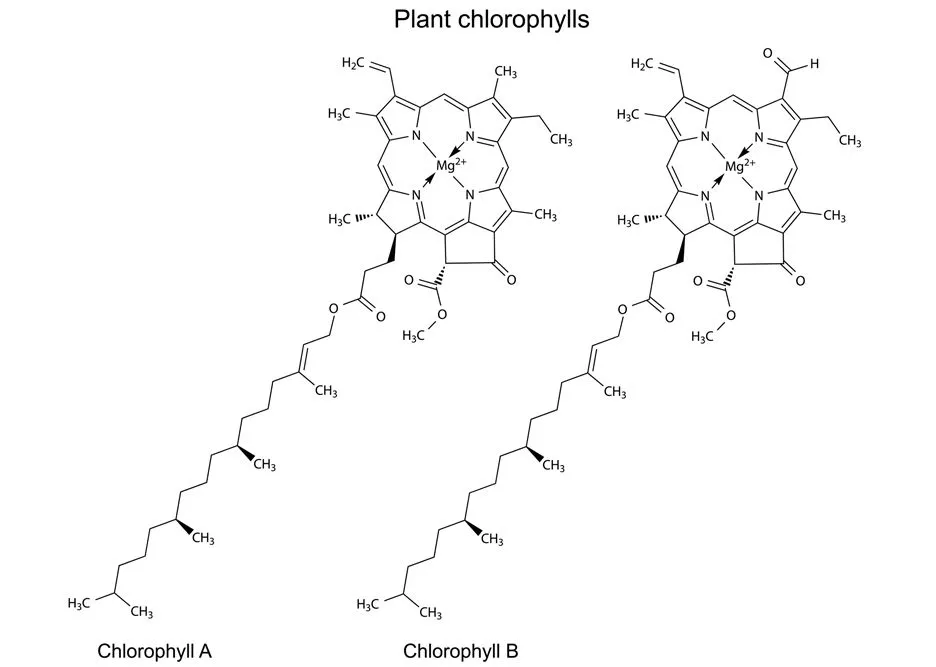
These absorption and reflection differences arise from structural and molecular differences in the chlorophyll molecules. These differences have thus far given us six broad categories of chlorophyll: a, b, c1, c2, d, f. Plants mostly have only two types of Chlorophyll in them: “a” and “b”. Chlorophyll a is teal-green in color, whereas Chlorophyll b is yellow-green in color. A combination of these two types of chlorophyll makes them into a shade of green.
Why Do Different Plants Have Different Shades Of Green?
However, these aren’t the only light-harvesting pigments present in leaves. To a much lesser degree, pigments that color orange, red, and yellow are also present in leaves, but they play a secondary role to chlorophyll. Carotenoids and xanthophyll are only two others of the numerous pigments within leaves that capture the energy of the sun. Carotenoids give vibrant orange and yellow hues. They are the pigments that give carrots their iconic orange. Xanthophylls are the reason sunflowers have such sunny yellow petals, as well as reds and oranges of various other fruits and vegetables.
The different concentrations of these pigment will dictate the color of green in the plants around us. The different types and amounts of pigment in different species of plant can reflect their evolutionary roots and reveal information about the plant’s habitat, its nutritional status and needs, and its age.
The Habitat Influences The Color Of A Plant
It’s all about survival. Where a plant grows and thrives can indicate a lot about its photosynthetic pigment concentrations. There are several factors that play a role here.
Light, its intensity and quality, are factors that affect the concentration of different chlorophylls, especially the ratio of chlorophyll a to chlorophyll b.
Plants that get abundant sunlight have less overall chlorophyll concentration and higher amounts of chlorophyll a than chlorophyll b. Plants that grow under shade, like those in densely forested areas, have a high overall chlorophyll concentration, but have more chlorophyll b than chlorophyll a.
Chlorophylls are excellent light harvesters, but too much light can be a bad thing. In sun plants, the excess and intense sunlight can harm the chlorophyll in its leaves. With the abundance of sunlight, less chlorophyll is enough, and to protect the chlorophyll in the leaves, these plants often have red pigmented regions. These red pigments serve to shade the chlorophyll and allow them to regenerate and utilize the sun’s energy. (Source)

For shade plants, the sun’s rays are a coveted commodity. To use the little they get as efficiently as possible, they make their light-harvesting complexes—the protein structures within which the chlorophyll are present—significantly larger. This means they can have more chlorophyll b pigments to absorb light, yet the amount of chlorophyll a changes very little, since they are mainly present at the center of the LHCs, and since the number of LHCs do not change, their concentration doesn’t increase either. Increasing chlorophyll b is important, since light under shaded canopies is filtered, and there is less red light available, which is the wavelength at which chlorophyll a absorbs best. Chlorophyll b, on the other hand, absorbs light at blue wavelengths more efficiently.
Some plants have thin leaves, but some have very thick leaves. Thicker leaves are either juicy (also called succulents) as in a cactus, or non-juicy (also called dicotyledons) as in Eucalyptus leaves. The leaves of dicotyledons are thick and non-juicy, as well as darker green, because they have dense chloroplasts that highly absorb sunlight, but have low reflectance. Succulent leaves tend to be lighter, as their cells are quite watery, so the concentration of chlorophyll on the surface is lower.
The upper part of a leaf is darker, owing to the high concentrations of chloroplasts present, as compared to the lighter bottom part.
Age Of The Leaf Influences Its Green Color
Young leaves have a lighter shade of green than matured and old leaves. Young leaves have yet to develop a fully functioning cell wall, and they can’t achieve photosynthesis as efficiently, both of which contribute to a lighter shade. As the leaves mature, they become darker green. Many leaves even turn yellow or orange before death, resulting in beautiful fall foliage in various parts of the world.
Deficiencies In Plants Influences The Green Color
Yes, we are not alone in this… even plants get nutrient deficiencies! Nutrient deficiencies such as that of nitrogen, low magnesium and iron (also called Chlorosis) can make the plant go yellow or yellowish-green. This happens because these chemical components are necessary for making chlorophyll.

Now that we know about the various shades of green in a garden, let’s play a game! You already know everything there is to know. Look around you and observe as many leaves as you can and start categorizing those plants according to their age!
References (click to expand)
- On the Hidden Colors in Leaves - Appalachian State Biology. Appalachian State University
- Photosynthetic Pigments - UCMP Berkeley. The University of California Museum of Paleontology
- Rodríguez-Castañeda, G. (2012, October 16). The world and its shades of green: a meta-analysis on trophic cascades across temperature and precipitation gradients. (M. Sykes, Ed.), Global Ecology and Biogeography. Wiley.
- Daughtry, C. (2000, November). Estimating Corn Leaf Chlorophyll Concentration from Leaf and Canopy Reflectance. Remote Sensing of Environment. Elsevier BV.
- Ruban, A. V. (2009, January). Plants in light. Communicative & Integrative Biology. Informa UK Limited.
- Mathur, S., Jain, L., & Jajoo, A. (2018, March 1). Photosynthetic efficiency in sun and shade plants. Photosynthetica. Institute of Experimental Botany.












