Table of Contents (click to expand)
Dielectrics are used in capacitors in order to increase the capacitance. This is because dielectrics increase the ability of the medium between the plates to resist ionization, which in turn increases the capacitance.
Dielectrics are basically insulators, materials that are poor conductors of electric current. Unlike the free electrons in a conductor, its electrons are tethered to its atoms. Consequently, no current can flow through it.
Such a material has no place in conductive devices, unless it is used to insulate itself, of course. However, if you think that dielectrics are despised by engineers, you are terribly mistaken. In fact, dielectrics are as ubiquitous as transistors. Between every capacitor is sandwiched a dielectric, the same capacitors without which your touchscreen would merely be a sheet of glass. But how does an insulator enhance the efficacy of a capacitor?

First we need to understand how a capacitor works.
Recommended Video for you:
The Capacitor
A capacitor is a device that consists of two parallel metallic plates placed extremely close to one another. The primary objective of a capacitor is to store charge. The charge can later be released to drive other circuits. This property renders it very useful in devices such as inverters. However, before releasing charge, it must first acquire it.
A capacitor is fed charge by connecting its plates to the terminals of a battery. Now, because metals are a sea of free electrons, when the electrons emanating from the negative terminal reach the metal, they violently repel the electrons on its surface. The repulsive force neutralizes the force exerted on the electrons by the battery, deterring them from accumulating on the plate.
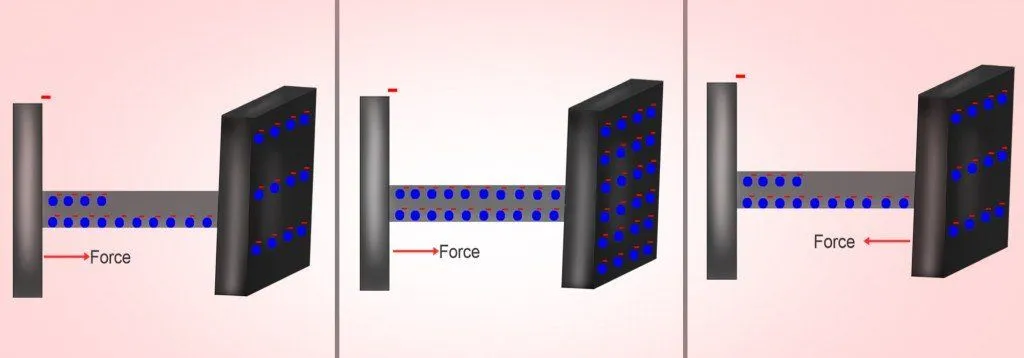
The extra electrons could be accommodated if they were somehow attracted by a force greater than the force of repulsion. This is achieved by placing another metal plate parallel to it. The parallel plate is connected to the positive end of the battery. At that point, the positive terminal attracts the electrons from the plate to which it is connected, rendering it positively charged.
This positively charged plate will now provide the force of attraction we desired, meaning that it will attract the extra electrons transmitted by the negative terminal. In this way, the capacitor will store charge. This charge on the plate can be used to drive another circuit in the absence of a battery by simply connecting the wires to the negative and positive plates as they typically are to the two terminals of a battery.
Why Dielectrics Enhance Capacitance
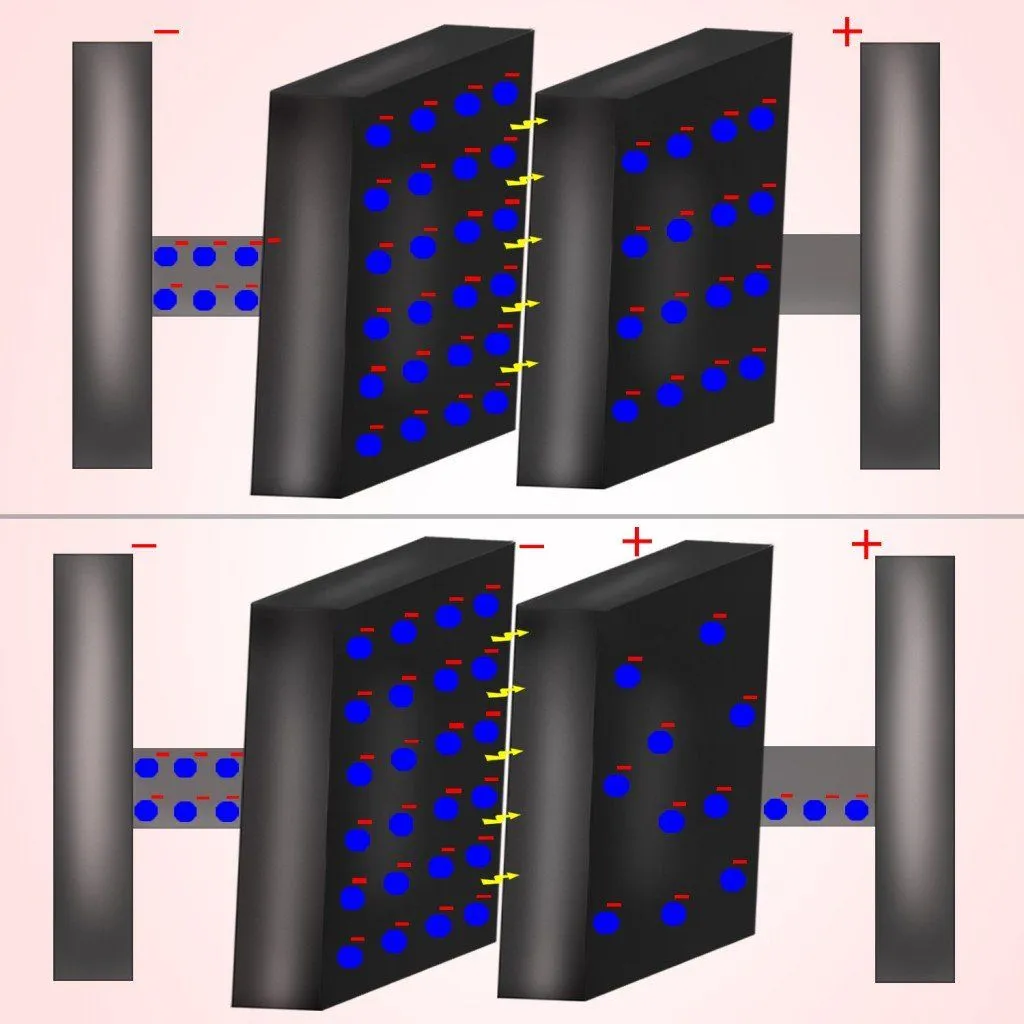
Even though atoms in a dielectric cannot be ionized to generate a current, they can certainly be polarized. When one brings a negatively charged object towards a dielectric, the electrons in its atoms are repelled by it. This accrues a net positive charge on the side of the dielectric facing the object and, consequently, a net negative charge on the opposite side.
Now, because an army of positive charges faces the negative plate, the electrons on the plate are now bound to the plate even more tightly. Also, because the second plate faces the electrons, the electrons on the plate are now repelled with greater efficacy. This will cause the first plate to accrue an even greater number of electrons, thus increasing the overall capacitance!
Capacitance is given by the ratio of the plates’ cross-sectional area and the distance between them. Capacitance will increase if we increase the cross-section of the plate for the obvious reason that a larger plate can accommodate more charges. Capacitance decreases with an increase in the distance between the plates for the simple reason that an increased distance weakens the attractive forces that lure and bind the electrons to the second plate.
However, we have just found that capacitance is also a direct function of the ability of the medium between the plates to resist ionization. The measure of this ability is given by its permittivity. Capacitance is therefore equal to the ratio of the plates’ cross-section and the distance between them, multiplied by the permittivity of the medium between them.
In conclusion, a good dielectric is not merely an insulator, but a material that refuses to ionize at any cost. They also ensure that the plates are always separated, thereby averting the possibility of a short. An even better dielectric is also robust and capable of working at higher temperatures.