Table of Contents (click to expand)
The answer to this question is yes, but only if you manage to eat 10,000,000 bananas in a rather concentrated period. If you did that, you would definitely die of radiation poisoning.
Sadly, the answer to this question is yes…. but only if you manage to eat 10,000,000 bananas in a rather concentrated period. If you did that, you would definitely die of radiation poisoning.
Luckily, one cannot eat such a massive quantity of bananas, considering that each banana, on average, amounts to 110 calories. Even so, this doesn’t change the fact that bananas do give off radiation. The secret to their radioactivity lies in the composition of the fruit itself. One medium ripe banana, on average, provides 1 gram of protein, 28 grams of carbohydrates, 15 grams of natural sugars, 3 grams of fibre and 450 mg of potassium. This all seems okay, just like a healthy fruit should be… so how can this give you radiation poisoning?
The answer lies in those small amounts of potassium present in this popular yellow fruit.

Recommended Video for you:
Is Potassium Radioactive?
Certain elements in nature consist of both stable and unstable (radioactive) atoms. Potassium is one such element. K-40 is the radioactive potassium isotope (elements with the same number of protons, but a different number of neutrons in the atoms). This is a naturally occurring isotope of potassium and is present in all plant and animal tissues. A banana contains both stable and unstable isotopes of potassium, with K-40 representing around 0.012% of the total potassium content in the fruit. Therefore, even if it’s just in small amounts, bananas are radioactive by nature.
K-40
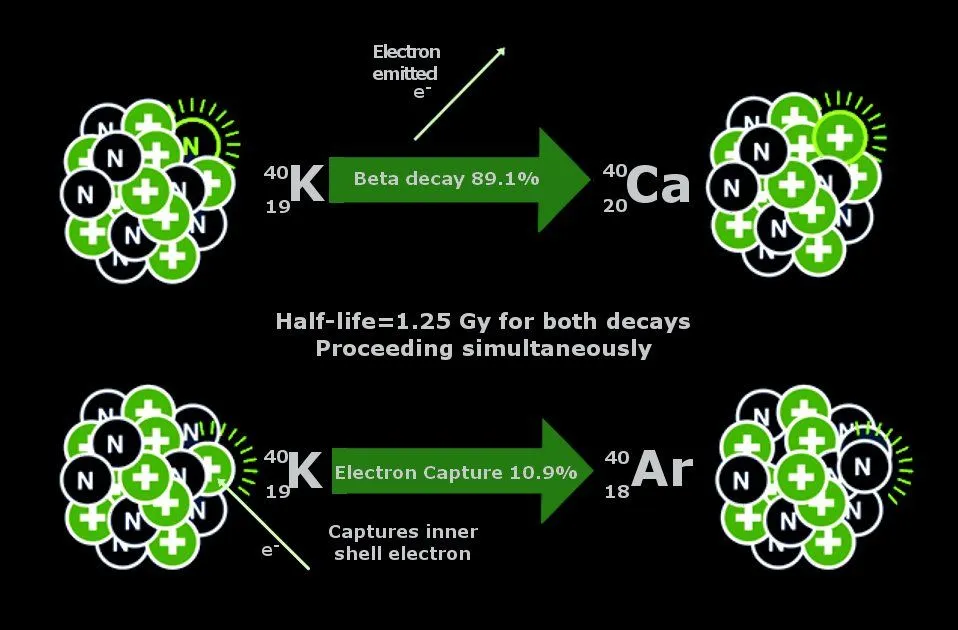
As the name suggests, this isotope of potassium has an atomic mass of 40, i.e., 19 protons and 21 neutrons in the nucleus of each atom, but what do these numbers tell us?
These are the numbers that determine the stability of these elements—and our fate upon being exposed to them. The ratio of the number of protons to the number of neutrons determines the stability of the atom. A ratio greater than 1 indicates instability—and the radioactivity of that element. Hence, K-40 is slightly radioactive. The very slow decay of potassium 40 into argon is highly useful for dating rocks, such as those that originate from lava, with age between a million and a billion years. The decaying of potassium into argon produces a gaseous atom that becomes trapped at the time of the crystallisation of lava. The atom can only escape when the lava is still liquid, but not after it solidifies. At that moment, the rock contains a certain amount of potassium, but no argon. Over time, as the potassium 40 disintegrates, the gaseous argon atoms slowly accumulate in the lava, where they get trapped. Measuring the amount of argon 40 formed since the solidification of the lava allows for an accurate measurement of the rock’s age.
What Does Radiation Poisoning Do, To The Human Body?
Not to burst your bubble, but radiation exposure to the body does not give you superpowers. Unlike our favourite superheroes, who were either lab rats at military weapon research labs or underwent genetic mutation after getting exposed to space rock radiation, in reality, humans undergo very different transformations. Depending on the duration of exposure and the strength of radiation, the symptoms vary widely. Dizziness, fatigue, hair loss, skin problems, fever and headaches are just the start. Due to the damage, it causes to the body cells, your overall immunity decreases, opening the gateway for a plethora of infections. The untimely intervention of doctors might lead to death in extreme cases.
Now that you know what radiation poisoning can do to your body, it’s worth mentioning that bananas are a radioactive food item. There are dozens of such naturally occurring food items that we ingest, almost on a daily basis, completely unaware of their radioactive nature. Potatoes, sunflower seeds, various nuts and kidney beans are just a few of them. Among these, Brazil nuts are the world’s most radioactive food, but what you probably don’t realise is that every type of food has a small amount of radioactive substances in it. The most common radioactive atoms present in food are potassium 40 (K-40), radium 226 (Ra-226) and uranium 238 (U-238).
Should We Be Concerned?
Luckily, our body maintains proper levels of potassium by flushing out the excess through urination and sweat. Also, the quantity of K-40 in a banana is too small to concern yourself with. The same can be concluded for other radioactive food items. How do these radioactive elements end up in our food? They come from the soil in which the foods are grown! Thus, every food item will have varying amounts of these naturally occurring radioactive elements.
In certain scenarios, the spillage of radioactive materials from industries into the soil leads to the contamination of food with excess amounts of radioactive material. This is a separate case altogether, one in which our not so harmful bananas could actually pose a danger to the consumer. Thus, it is of utmost importance that radioactive waste is handled correctly and disposed off , using proper measures.

Conclusion
We often fail to realise how exposed we are to these forms of radiation, yet we generally remain unaffected by them. We are not entirely protected, but our knowledge of radioactive substances is critical, as they have found their way into a wide range of medical research and treatments.
About one-third of all patients admitted to hospitals are diagnosed or treated using radiation or radioactive materials. The radioactive materials used are called radiopharmaceuticals. A small amount of radioactive material is injected, inhaled or swallowed. The material becomes concentrated in the area under study, where it emits photons. These photons can be seen by a device known as a gamma camera, which can help physicians find and identify tumours or view problems in an organ. Radioactive iodine is also used when imaging the thyroid gland.
As far as our banana is concerned, there is little to no risk of being affected by radiation exposure, but there is a risk of slipping on the peel that you threw on the ground, so pick up after yourself!
References (click to expand)
- Bananas | The Nutrition Source | Harvard T.H. Chan School of .... The Harvard T.H. Chan School of Public Health
- Potassium-40 - phi.nmsu.edu:80
- Radiation sickness - Symptoms and causes. The Mayo Clinic
- Definition of radiation poisoning - NCI Dictionary of Cancer Terms - www.cancer.gov
- Radioactive foods|Scientific India Magazine - www.scind.org:80
- Naturally Occurring Radioactive Materials NORM. World Nuclear Association
- Backgrounder On Medical Use Of Radioactive Materials. The Nuclear Regulatory Commission












