Table of Contents (click to expand)
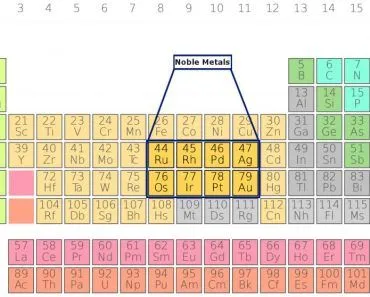
Aqua regia is a very strong acid formed by the combination of nitric acid and hydrochloric acid. The term ‘aqua regia’ is Latin for ‘royal water’. It has been popular for its ability to dissolve gold, palladium and platinum, all of which don’t react easily with chemicals.
Aqua regia is a very strong acid formed by the combination of concentrated nitric acid and concentrated hydrochloric acid, both of which are strong acids. It is generally used to remove metals like gold and platinum from substances, especially in microelectronics and micro-fabrications labs.
After dissolving the gold medals, he placed the jar on a shelf that held numerous other jars and bottles, all of which looked similar (at least to the untrained eyes of Nazi soldiers, most of which were unlikely to have a degree in Chemistry).
All those bottles, including the jar that contained the dissolved gold medals of the two Hungarian scientists, were left untouched, and Mr. De Hevesy and his rather unusual act of heroism became immortal in the corridors of chemistry labs all over the world.