Table of Contents (click to expand)
Rancidity occurs when fats and oils are exposed to air, light, moisture, and bacteria, leading to hydrolysis or oxidation. This renders food items unfit for consumption.
Have you ever wondered why a bag of chips is always half full? The reason has to do with a concept called rancidity. No food item can remain fresh for an extended period. They become spoiled due to various chemical reactions, and rancidity is one of them.
When food items containing fats and oils are exposed to air or moisture, they become spoiled. To prevent chip spoilage, manufacturers flush nitrogen gas into the chip bags. Nitrogen is used in bags of chips because it does not react with fats and oils.
Recommended Video for you:
How A Substance Turns Rancid?
Rancidity describes the spoilage of food products containing oils and fats. Most edible fats and oils are made up of triglycerides—three fatty acid chains attached to a glycerol backbone. A fatty acid itself is a long aliphatic chain of carbon atoms ending in a carboxylic acid (–COOH) group. Saturated fatty acids have only single bonds between their carbon atoms, while unsaturated fatty acids contain one or more carbon–carbon double bonds, which is precisely where they are most vulnerable to attack by oxygen.
Rancidity Reactions Typically Occur In Three Steps:
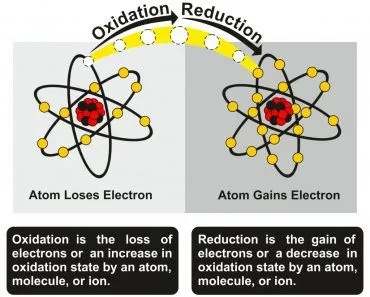
1. Initiation reaction, triggered by external factors such as heat, light, or trace metal catalysts, removes a hydrogen atom from a fatty acid (RH) and creates a carbon-centred lipid radical. By definition, a radical is an atom or molecule that carries an unpaired electron, which makes it highly reactive.
RH ———> R• + H•
2. Propagation reaction, where the lipid radical reacts almost instantly with molecular oxygen to form a peroxyl radical (ROO•). The peroxyl radical then steals a hydrogen atom from a neighbouring fatty acid, producing a stable lipid hydroperoxide (ROOH) and a brand-new lipid radical—keeping the chain reaction going.
R• + O2 ———> ROO•
ROO• + RH ———> ROOH + R•
3. Termination reaction, where two radicals collide and combine to form non-radical products, ending the chain.
ROO• + ROO• ———> non-radical end products (+ O2)
The hydroperoxides (ROOH) formed during propagation are unstable and break down into smaller aldehydes, ketones, and short-chain acids. These secondary products—not the hydroperoxides themselves—are what give rancid foods their characteristic stale, soapy, or paint-like odour and taste. Rancidification can also destroy fat-soluble vitamins such as A and E in the food.
Types Of Rancidity
Rancidity can be classified into two major types: oxidative rancidity and hydrolytic rancidity.
Oxidative Rancidity
Oxidative rancidity is a reaction that damages a food substance by causing oxygen damage. This process interrupts and damages the natural oil structure in a way that can alter its color, odor, and taste. Furthermore, it forms toxic compounds like peroxides, which can destroy vitamins A and E in foods. The reaction also creates polymeric materials and oxidized sterols.
Hydrolytic Rancidity
Hydrolytic rancidity generally causes an unpleasant odor. This is due to the release of free fatty acids from glycerides.
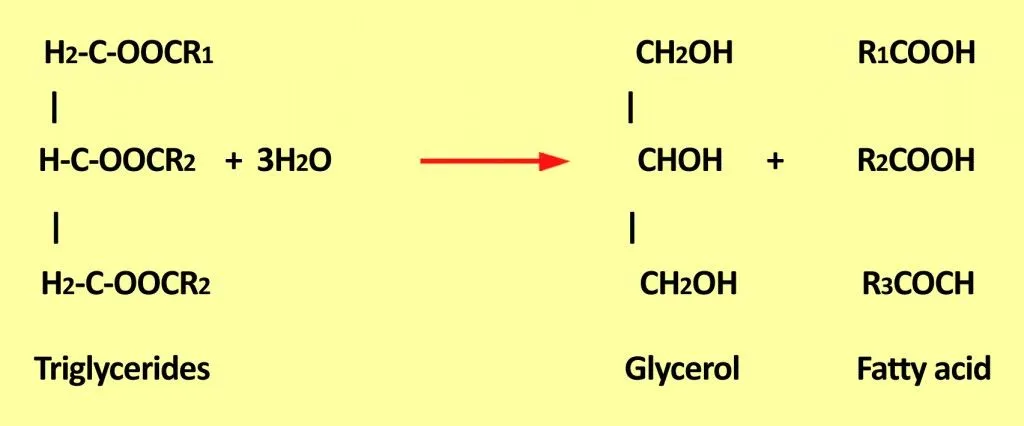
Triglycerides are a combination of three fatty acids that are generally present in oil foods. When they react with water, they produce glycerol and release free fatty acids unsuitable for consumption.
These fatty acids may further undergo oxidative rancidification, forming toxic compounds.
The classic example is butter going rancid. About 4% of fresh butter is a triglyceride of butyric acid; once exposed to moisture and lipase enzymes from microbes, the triglycerides hydrolyse and free butyric acid is released, giving spoiled butter its sharp, sour, “baby-vomit” smell. The same mechanism is responsible for the off-taste of stale milk, ghee, and coconut oil.
Common Examples Of Rancidity
Rancidity shows up in nearly every fat- or oil-rich food we eat. Here are five everyday examples:
- Stale potato chips — when an opened bag is left out, the frying oil on the chips reacts with oxygen and develops a cardboard-like or paint-like smell. This is why unopened bags are flushed with nitrogen gas, an inert gas that does not react with the oil and slows oxidation.
- Spoiled butter and ghee — moisture and lipase break the triglycerides into glycerol and free butyric acid (hydrolytic rancidity), producing a sharp, sour odour.
- Old cooking oil — repeatedly used or improperly stored vegetable oil turns dark, smells “off”, and produces foam while frying because of accumulated peroxides and aldehydes.
- Stale nuts and seeds — almonds, walnuts, and flaxseeds are rich in unsaturated fats and quickly develop a bitter, sharp taste once their oils oxidise.
- Fish and dried meat — the polyunsaturated fats in fish oxidise rapidly, producing the characteristic “fishy” smell associated with spoilage.
Factors That Affect Rancidity
- Oxygen: Exposure to oxygen is the primary cause of rancidity. Oxygen is more soluble in fats, which leads to oxidation and food damage by releasing free radicals.
- Microorganisms: Several microorganisms release an enzyme called lipase that leads to the hydrolysis of lipids. This enzyme further acts on oils and fats. Lipase requires a certain pH and temperature for it to react.
- Physical factors: Factors like temperature, heat, and light also play a significant role in rancidification. Heat and light are the major sources for the production of free radicals. Heat and light accelerate the process of oxidation.
How To Prevent Rancidity?
To retain the desirable qualities of food products, preventing them from becoming rancid is essential. One of the easiest ways to do this is by keeping them away from direct contact with light and air. You can store them in airtight containers for this purpose.
Adding antioxidants is also an effective way to prevent auto-oxidation in foods that contain fats and oils. Antioxidants can be either natural or synthetic. Natural antioxidants include vitamin C, vitamin E, flavonoids, and polyphenols. Sequestering agents such as EDTA also prevent or slow down oxidation and, therefore, can help effectively prevent rancidity.
Last Updated By: Ashish Tiwari
References (click to expand)
- Oxidation of food grade oils - Oils, fats and more.
- Esfarjani, F., Khoshtinat, K., Zargaraan, A., Mohammadi‐Nasrabadi, F., Salmani, Y., Saghafi, Z., … Bahmaei, M. (2019, June 6). Evaluating the rancidity and quality of discarded oils in fast food restaurants. Food Science & Nutrition. Wiley.
- Vegetable oil oxidation: Mechanisms, impacts on quality, and approaches to enhance shelf life - PMC.
- Rancidification - Wikipedia.