The Tyndall Effect is the phenomenon of the scattering of light by colloidal solutions and suspensions. It is responsible for our blue skies, the scattering of light in fog and many other fascinating events that we easily take for granted!
After beginning its 8-minute journey to reach the surface of our planet, sunlight goes through many physical spaces that change it in certain aspects. Aside from the run’s rays, the light generated and emitted by electronic instruments is also subjected to many changes in our atmosphere.
For instance, the color of the sky changes as light moves through the atmosphere. Driving on a cold blue winter night full of fog, people often use the fog lights present in their vehicles. These instruments release rays that illuminate the path ahead, regardless of the amount of fog present.
The mysteries of light mentioned above and many more can be unravelled by studying the simple concept of the Tyndall Effect. However, to understand this phenomenon, one should first be acquainted with the process of scattering light.
Recommended Video for you:
What Is The Scattering Of Light?
The scattering of light implies distributing it in all directions. When a parallel beam of rays strikes a particle of appropriate size (smaller than its own wavelength), the particle absorbs the rays and emits (or releases) them in all the directions, except the direction by which the rays arrived; this entire phenomena is known as the scattering of light. The size of the particle, as mentioned earlier, is a key aspect of this phenomena, as particles of all sizes cannot scatter light of all wavelengths.
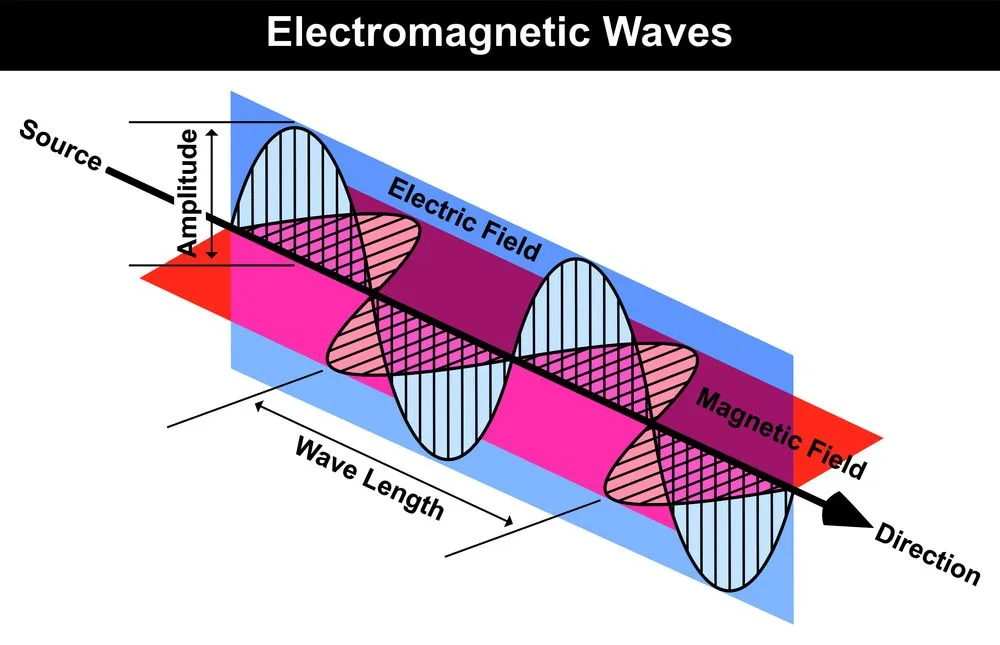
Light beams are nothing but electromagnetic waves (radiation). A ray of light has many properties that define its characteristics. Two of the most important of these are its frequency and wavelength. The size of light waves is measured as the wavelength, which is the distance between any two corresponding points on successive waves (we typically measure this as the distance between two continuous peaks or crests of a wave). The frequency of a beam, on the other hand, is the number of waves that pass a particular point in space every second.
What Is The Tyndall Effect?
The Tyndall Effect is the phenomenon of the scattering of light by the particles present in a colloid or very fine suspension. To be classified as a colloidal solution, a material must have particles with dimensions (length, width, thickness) in the range of 1-1000 nanometers. Suspensions are heterogeneous mixtures composed of solid particles that do not dissolve in the liquid or gas present. The size of the particles can range from 10 to 1000 micrometers. The only difference betweena colloid and a suspension is the size of the particles.
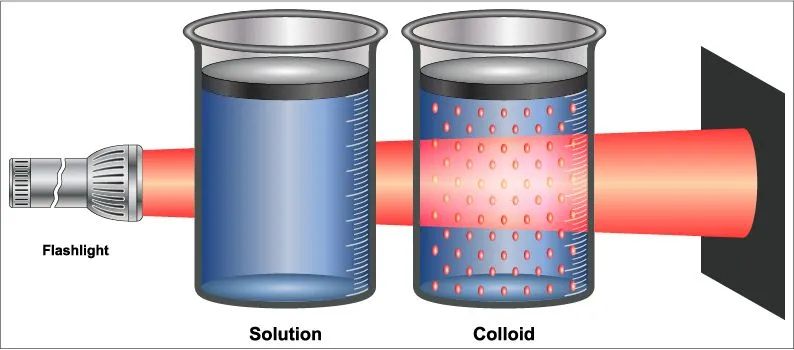
Some common examples of colloids used in our everyday life are whipped cream, mayonnaise, butter, milk, gelatin and paper. Some examples of suspensions include salt water, sugar water, oil paints and muddy water.
The light emitted from a particle after the scattering occurs appears to be of different colors, depending on its wavelength. The reason behind this selective scattering is the relationship between the intensity of the light being scattered and its wavelength. The amount of light scattered is inversely proportional to the fourth power of its wavelength. This means that shorter wavelengths of light (such as violet or blue) are scattered far more than longer wavelengths (such as red or yellow).
The Tyndall Effect On A Molecular Level
When light strikes a molecule that is smaller than its wavelength, it’s absorbed by the particle. The electric fields of this ray temporarily polarize the molecule by pushing the electrons in one direction. This leads to the formation of a weak net electrical charge—positive on one side and negative on the other—which is known as a dipole moment. As light propagates inside the molecule, the direction of the dipole keeps changing, and as it re-interacts with the electric fields of the rays, the molecule emits them in all directions.
How Does The Tyndall Effect Lead To Blue Skies?
Our sky (the one closer to Earth’s surface and visible to us) is composed of gases like oxygen, nitrogen and carbon dioxide. The molecules of these gases have diameters that are smaller than the incident white radiations of the sun, which are composed of waves of different colors (wavelengths). As a result, when sunlight strikes these molecules, the shorter waves of violet and blue are scattered the most, giving our sky its signature bluish color. Given that the cells of our eyes are more susceptible to blue color over violet, the sky appears blue to us.
Similarly, during sunrise and sunset, light has to travel the longest to reach our eyes. In such a situation, blue light is completely scattered, leaving behind only waves of red and yellow. Thus, the sky appears orange or red during sunrise and sunset. In foggy weather, light rays strike the particles of fog (a colloidal solution) and are scattered, thus increasing visibility, even at night.

A Final Word
When John Tyndall discovered the phenomenon of scattering light from colloids, he probably didn’t think that it would lead to the foundation of a new branch of science—Spectroscopy. This area of study deals with detecting the properties of an unknown substance by analyzing the spectrum of light emitted by it.
Similarly, many small discoveries have led to the beginning of something much larger that has helped humanity’s quest for understanding this universe. In other words, our imagination should not be limited by the scale of an idea. Keep learning and keep thinking—these are two of our greatest gifts!