Table of Contents (click to expand)
Water is less dense in its frozen form, ice, than it is in its liquid form. This is because the molecules in ice are further apart than the molecules in liquid water. The molecules in ice are held further apart by the hydrogen bonds between the oxygen and hydrogen atoms.
From giant icebergs to tiny cubes, ice – the frozen form of water – always floats on its liquid form. Isn’t that weird?
We’re not the only ones who think it’s unusual; the entire world finds it rather surprising that a solid should float on its liquid form. Do a quick Google search and you’ll find dozens of pages discussing this queer tendency of ice.
As it turns out, like everything else, there is a scientific reason behind this phenomenon. But first off, let’s be clear about what makes stuff sink or float.
The singular rule of thumb, when it comes to the ability of an object to float in water (or any other liquid), concerns the density of the object in question. Have you ever heard of Archimedes’ Principle?
Archimedes’ Principle states that for an object to float on water, it must displace an equal amount of water. In other words, you can say that the fate of an object in a body of water is decided by Archimedes’ Principle.
It’s common knowledge that solid objects have more density than their liquid counterparts. Chemically, this makes sense too. Molecules are more closely bound with one another in a well-defined manner in a solid, which makes them rigid and gives them more weight. All common substances that we see and observe in daily life follow this basic principle: solid objects are denser and have more weight than liquids.
Given that, why does ice – which is a solid – float on water? Shouldn’t it sink, as a solid, and according to general convention, also have more density?
Recommended Video for you:
A Little Water Info
Water is a wonderful liquid, and full of unusual behaviors and chemical structures, which is why it presents an interesting exception to the general behavior of solids floating over their liquid forms.
If you keep cooling a liquid, its density continues to increase until it becomes solid, where it attains maximum density. However, in the case of water, this trend is slightly different, which is the root cause of this whole discussion.
Water’s density increases as you continue to cool it; but unlike most liquids, which reach their highest density when they freeze, water achieves maximum density at about 3.98 °C (39.16 °F, often rounded to 4 °C) — roughly 999.97 kg/m³.
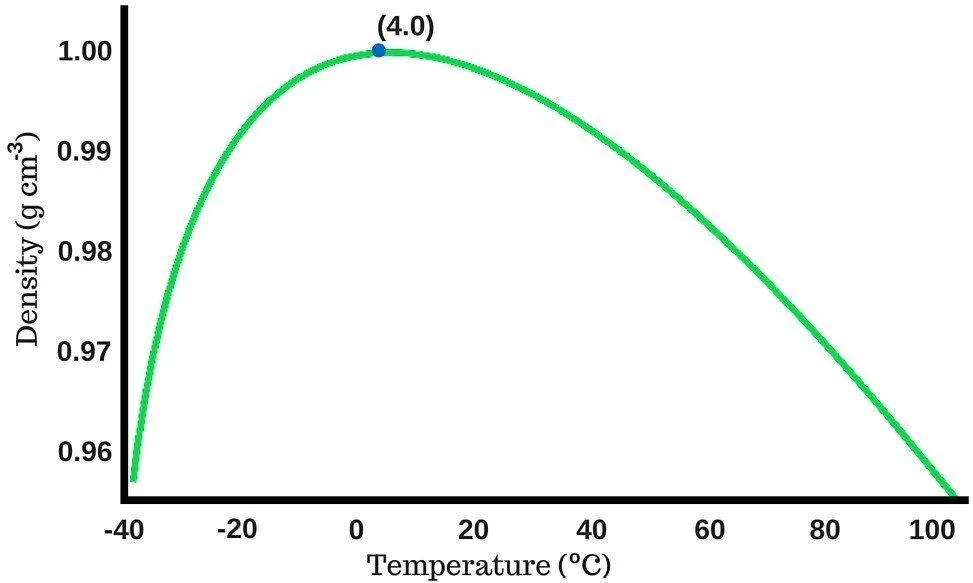
If you continue to cool water past 4 °C, its density actually drops slightly (you can see this in the graph). Just above freezing, liquid water sits at ~999.84 kg/m³, but the moment it crystallises into ice the density falls sharply to about 917 kg/m³ — nearly an 8% drop, which is why ice cubes bob with most of their bulk underwater.
It turns out that ice has a lower density than water, and any object that has a lower density than the liquid form on which it’s kept (in this case, water) will be able to float!
But Why Is The Density Of Ice Less Than Water?
To answer that, you’ll have to look at the chemical structure of water.
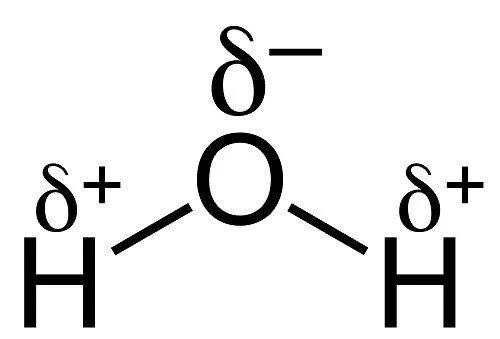
Each water molecule has a slightly negative oxygen atom and two slightly positive hydrogens (because oxygen is more electronegative). The negative oxygen of one molecule attracts a positive hydrogen of a neighbouring molecule — this intermolecular attraction is what we call a hydrogen bond. (It’s separate from the much stronger covalent O–H bond inside each molecule.)
When most liquids are cooled, the molecules pack more closely together, which is why the corresponding solid is usually denser than its liquid form. Water plays a different game. As it freezes, the hydrogen bonds between molecules lock into a fixed, open tetrahedral arrangement — each water molecule hydrogen-bonds to four neighbours, producing the hexagonal lattice of ice Ih with plenty of empty space inside. When ice melts, about 10–15% of those bonds snap, the lattice partially collapses, and the molecules can crowd in closer than they could in the rigid crystal. That’s the deep reason ice ends up less dense than the liquid it came from.













