Table of Contents (click to expand)
Heavy metals are toxic to living things and when they are present in the environment, they can wreak havoc on organisms. Some bacteria have come up with ways to deal with this toxicity.
Microbes are tiny living organisms that are omnipresent in our environment. From the soil and rocks to the oceans, from the highest mountains to the deepest trenches, and from the hottest hydrothermal vents to the coldest glaciers, no nook is free of microbes; they’re even present inside plants, animals, and human beings. They are so versatile that they can thrive everywhere and on everything.
However, there are certain prerequisites for microbes to grow with such ease and speed. These include nutrients that contain a carbon source, a nitrogen source, vitamins, and minerals. They also require water in the form of moisture, appropriate pH, amicable salt concentration, and appropriate oxygen conditions.

Recommended Video for you:
What Are Heavy Metals?
Heavy metals are inorganic elements that may prove toxic to living organisms when their concentration increases beyond the threshold limit of a particular environment.
Don’t get confused between minerals and metals; although many minerals are metals, all metals are not minerals.
Minerals are inorganic complexes with a definite structure and composition, both of which are vital to bacterial growth. By nature, they may either be metallic, such as potassium, magnesium, calcium, and iron, or non-metallic, such as phosphorous, sulfur, and chlorine. Minerals often play a biological role in the functioning of living organisms.

Most metals lack a biological role and may prove toxic to an organism when present beyond a certain threshold concentration. Some examples include lead, chromium, copper, cadmium, arsenic, zinc, mercury, and nickel.
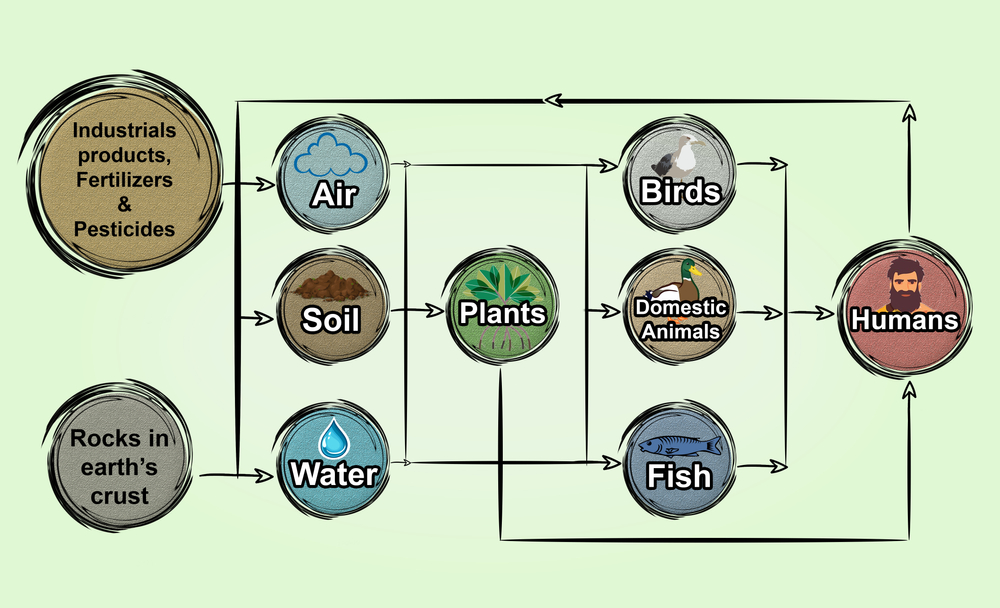
We get our minerals from the food that we consume, but what are the sources of heavy metals? Where do they come from?
The source of metal accumulation can be agricultural, industrial, or domestic. Agricultural sources include the use of inorganic fertilizers, pesticides, fungicides, and sewage waste, whereas industrial sources encompass refineries, metal ore extraction, coal extraction, petrochemical spillage, and thermal power. The domestic burning of biomass, organic and inorganic daily waste, used batteries, and used filters are also forms of domestic waste contributing to heavy metal accumulation.
These accumulated heavy metals persist in the soil much longer than organic contaminants. As soil is the major sink for heavy metal accumulation, life forms who call the soil home are put at risk. As soil harbors the most diverse microbial community and the highest microbial load, soil microbes are at greater risk of heavy metal contamination.
What Do Heavy Metals Do To Microbes?
Heavy metal accumulation in the soil poses more dangers to microbes than the accumulation of organic pollutants. The key reason is the leaching nature of the organic pollutants, which reduces their toxicity over time, while the heavy metals persist for a much longer time in the soil. Heavy metals can neither be destroyed nor broken down into smaller forms.
Heavy metals affect the soil microbial activity and the soil microbial community composition. Let’s briefly explore how this happens.
Effect Of Metals On Microbial Activity
Heavy metals hamper several microbial functions, such as the maintenance of soil structure, the formation of soil organic matter, the maintenance of biogeochemical cycles, and the decomposition of toxic compounds, which in the long run may threaten the function of the soil ecosystem.
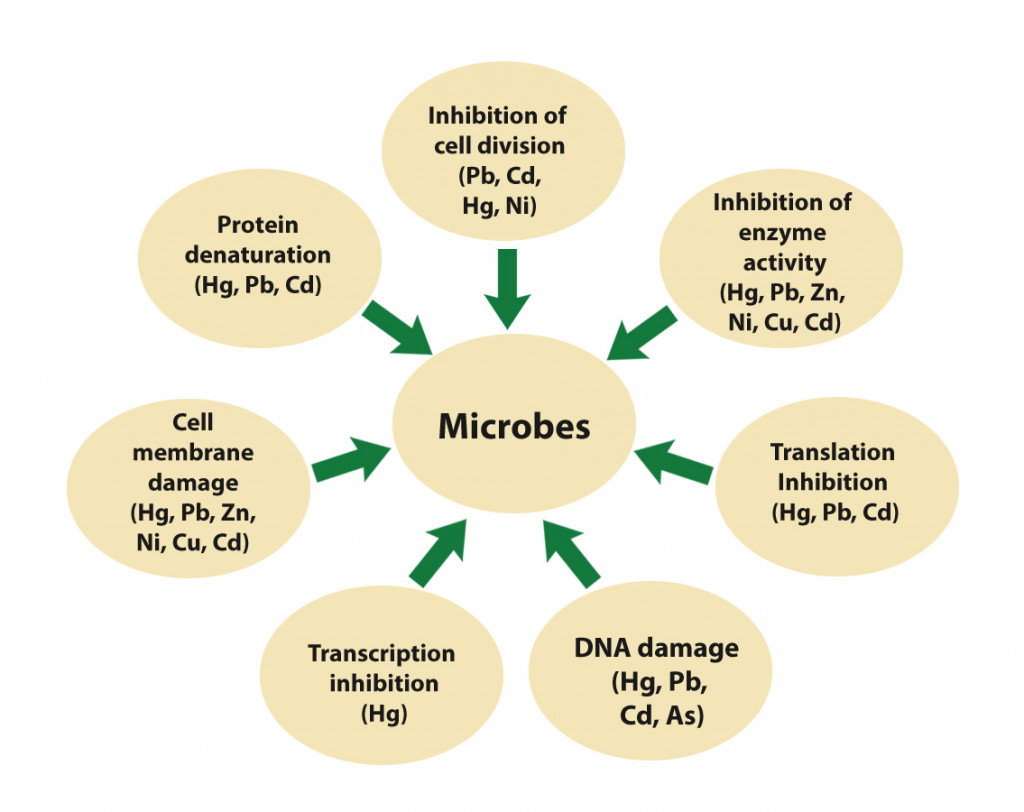
Let’s see which heavy metals affect what aspects of microbial metabolism.
Mercury, cadmium, and lead inhibit cell division, enzyme activity and translation, denature proteins, induce DNA damage, and damage cell membranes through ion imbalance. Ion imbalance occurs when the heavy metals displace the important minerals from the cell membrane and take their place instead. In addition, mercury also inhibits transcription, while arsenic induces DNA damage. Copper, nickel, and zinc inhibit enzyme activity and cause an ion imbalance. The activities of enzymes like superoxide dismutase, catalase, and ascorbate peroxidase are inhibited by heavy metals. They also contribute to the production of reactive oxygen species and induce oxidative damage.
Thus, heavy metals greatly influence the morphology, metabolism, growth, and reproduction of microbes.
Effect Of Metals On Microbial Community Composition
Heavy metals that have accumulated in the soil will alter the nutrient quality of the soil. If the nutrient quality deteriorates, the number of microbes thriving in the soil will also decrease. This reduction will restrict the release of CO2 in the soil, which will influence the microbial communities inhabiting the soil. This keeps repeating like a cycle.
Only those bacteria that can tolerate heavy metals, called metallotolerant bacteria, will be able to survive, which will eventually result in an alteration of the soil’s microbial community.
What Are The Mechanisms For Metal Tolerance In Microbes?
Some microbes have developed mechanisms to combat or tolerate the increased amounts of heavy metals in their environment.
There are five main mechanisms of metal tolerance.
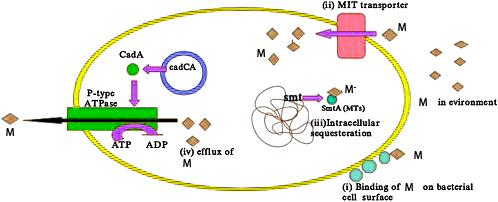
The first mechanism is the most straightforward: preventing the metal from entering the cell. This can be achieved in two ways. One way is to block the transporters (tunnels that allow molecules to move in and out of the cell) that are important for the transport of heavy metals from the outside of the cell to the inside. The second way is the extracellular production of slimy compounds to trap the metals and inhibit them from entering the cell.
If the microbes are unable to inhibit the uptake of heavy metals, they can try to kick the metals out of the cell. Even though this process is energy-consuming, it is feasible compared to the numerous negative effects the metals may impose on the microbes.
Microbes may try to form insoluble complexes with heavy metals, also termed as sequestration or accumulation, such that they don’t exert their deleterious effects. This may be done intracellularly or extracellularly, and the mechanism to achieve this varies from microbe to microbe.
Several microbes are even capable of using toxic heavy metals for the purpose of energy generation. This poses a dual advantage of rendering the metal non-toxic, while also gaining energy.
Can you believe how smart microbes are? They’ve not only evolved to exhibit mechanisms to combat metal toxicity, but even use the same toxic metals for energy generation!
Conclusion
The ever-increasing anthropogenic activities on our planet have resulted in the excessive accumulation of contaminants like heavy metals in the soil. Their persistent, toxic and non-biodegradable nature deteriorates not only the soil quality, but also its microbial richness. As microbes are more sensitive to environmental stress, they act as important indicators of soil quality. The constant exposure of heavy metals to the microbes has resulted in the evolution of microbes with adapted mechanisms to help them survive heavy metal stress.
References (click to expand)
- Salt - seafoodhaccp.cornell.edu:80
- Minerals & Elements. mineralseducationcoalition.org
- Chu, D. (2018, February). Effects of heavy metals on soil microbial community. IOP Conference Series: Earth and Environmental Science. IOP Publishing.
- Wuana, R. A., & Okieimen, F. E. (2011, October 24). Heavy Metals in Contaminated Soils: A Review of Sources, Chemistry, Risks and Best Available Strategies for Remediation. ISRN Ecology. Hindawi Limited.
- (PDF) [Mechanisms of bacteria resistance to heavy metals]. ResearchGate
- Microbes in heavy metal remediation - nopr.niscair.res.in
- Igiri, B. E., Okoduwa, S. I. R., Idoko, G. O., Akabuogu, E. P., Adeyi, A. O., & Ejiogu, I. K. (2018, September 27). Toxicity and Bioremediation of Heavy Metals Contaminated Ecosystem from Tannery Wastewater: A Review. Journal of Toxicology. Hindawi Limited.