Table of Contents (click to expand)
Candles work by using heat to produce light. In order to do that, they make use of a process called combustion. Combustion is basically the burning of a substance in the presence of oxygen, and involves the production of heat and light. Here, the fuel is wax, which is composed of complex carbon compounds. Wax burns in the presence of oxygen to produce carbon dioxide gas, which is colorless. The smoke that you see is because wax never burns in a completely clean way.
Eleanor Roosevelt once said, “It is better to light a candle than curse the darkness.”

Candles are not only an amazing lighting system, but they are also symbols of peace and serenity. Furthermore, candles are used for a variety of purposes; shouting “Boo” at someone with a candle held up to one’s face is a good way. Candle-lit dinners also don’t need any introduction, particularly for the romantics among us. Some people also use candles for their fragrance! Like I said, candles boast a variety of functions.
Now, let’s take a closer look at how a simple combination of a wick and some wax can bring light to the darkness.
Recommended Video for you:
The Parts: Wick And Wax
Wick: This is the little part of a candle that pokes out at the top. This is the part that burns and produces a flame. It is usually a string of braided cotton that holds the flame of a candle for a set period of time, depending on the amount of the wick that is used in the candle. This works by conveying the fuel (wax) to the candle, a phenomenon known as ‘capillary action.’ The candle wick greatly influences how a candle burns.

Also Read: Why Does A Candle Only Produce Smoke When It’s Extinguished?
Candle And Combustion
A candle uses heat to produce light. In order to do that, it makes use of a process called combustion. Combustion is basically the burning of a substance in the presence of oxygen, and involves the production of heat and light. Here, the fuel is wax, which is composed of complex carbon compounds. Wax burns in the presence of oxygen to produce carbon dioxide gas, which is colorless. The smoke that you see is because wax never burns in a completely clean way.
Also Read: What Determines The Color Of Flames?
The Process: What Happens When A Candle Burns?
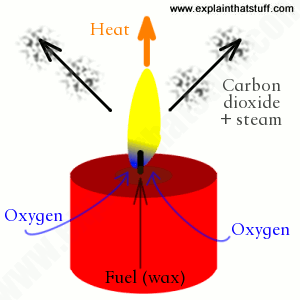 As soon as the wick is lit by some sort of fire-source (most commonly matchsticks), a chain of events begins. Wax, which is present on and near the wick, begins to melt. At that point, the wick absorbs the liquid wax (which has just melted) and pulls it upwards (due to capillary action). The heat of the flame vaporizes the fuel (molten wax), which in turn combines with oxygen in the air (combustion) to produce a constant flame.
As soon as the wick is lit by some sort of fire-source (most commonly matchsticks), a chain of events begins. Wax, which is present on and near the wick, begins to melt. At that point, the wick absorbs the liquid wax (which has just melted) and pulls it upwards (due to capillary action). The heat of the flame vaporizes the fuel (molten wax), which in turn combines with oxygen in the air (combustion) to produce a constant flame.
Congratulations! You’ve just lit a candle.
The flame you’ve just lit then melts the wax on and near the wick, and the whole cycle is repeated. This process goes on until there is a shortage (or cutoff) of fuel or oxygen, which causes the flame to extinguish.

The candle is such a simple tool for producing light, yet the science behind it is absolutely fascinating. Lighting a candle is one of those mysterious acts in life that we have become so used to over time that we cease to appreciate how cool the entire process is. Next time you need to light up the darkness during a power outage, take the time to properly appreciate just how much candles can do!












