Table of Contents (click to expand)
When you cook an egg, you’re changing the structure of the proteins inside it. Heating the egg makes the proteins uncoil and come together, while whipping or frying it makes them form a network that doesn’t let light through. This makes the egg whites harder and the yolk firmer.
Most mornings when I start preparing my breakfast, which typically consists of an egg, either boiled or fried, and toast, I never considered why I’m doing what I’m doing. I just break the egg in the pan and wait for the translucent egg whites to turn opaque. I never used to give a second thought that every delicious morsel that eventually makes its way into my mouth is a coagulated protein that has been beaten, fried, whipped or poached to be transformed into such a delicious treat.
I never asked a seemingly obvious question: why am I heating the eggs? Why not just eat them raw?
Recommended Video for you:
Heat ‘Em Up!

First of all, eating raw eggs is not a good idea, considering that you could become very ill by catching a food-borne disease (salmonella, to name a major one).
We heat an egg before eating it in order to cook it fully. How does subjecting it to heat help this process? Well, do you see the translucent egg whites in the picture above? They actually contain loads and loads of proteins, and these proteins are all tangled up, creating that viscous quality. Proteins are made of amino acids, which are bonded to each other with various weak bonds.
Therefore, when you crack your egg into some butter (or olive oil for those who want to eat healthy), do you notice how it starts to turn more opaque? This change is due to the tangled proteins getting uncoiled when their bonds break in the heat.
These uncoiled proteins then bond with other uncoiled proteins and form a large interconnected network that doesn’t allow light to pass through it. Voila! You have hardened egg whites.
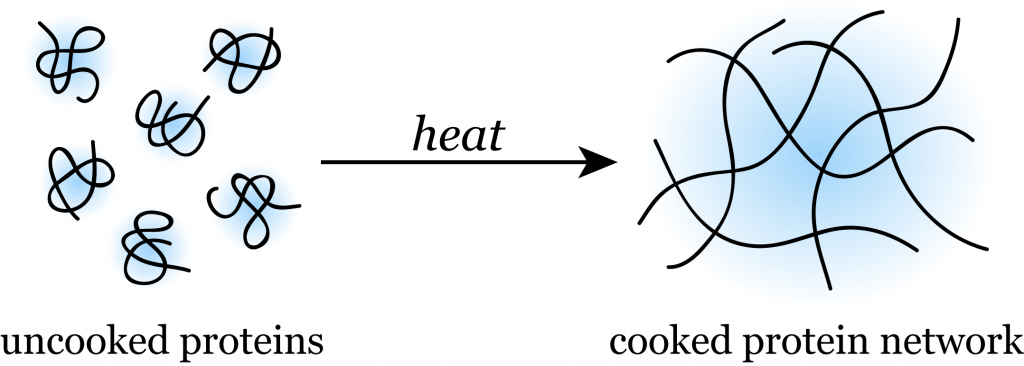
Don’t heat the eggs too much, though, or else you’ll get rubbery eggs and your breakfast will be ruined!
Beat ‘Em Up!

Are you making a meringue from scratch? Awesome! And what are you doing there? Beating your eggs, I see. What does that do? Why does an egg have to beaten and fluffy before you add any more ingredients to it? Well, in this case, what you need to know is that an egg is basically 90% water and 10% protein, so it’s basically a water-protein mix.
You have your whisk in hand and your eggs are ready for a good beating. As you start beating them, you’re incorporating air into that water-protein mixture. This also makes the proteins uncurl, just like when you apply heat. But why air? Well, all the amino acids that make up the protein have a water-loving hydrophilic part on the outside, closer to the water, and a water-hating hydrophobic part at the center, which is the coiled state.
So, when you start whipping it, those proteins start to uncoil. The hydrophobic part stays exposed to the air, away from the water, while the hydrophilic part is immersed in the water.
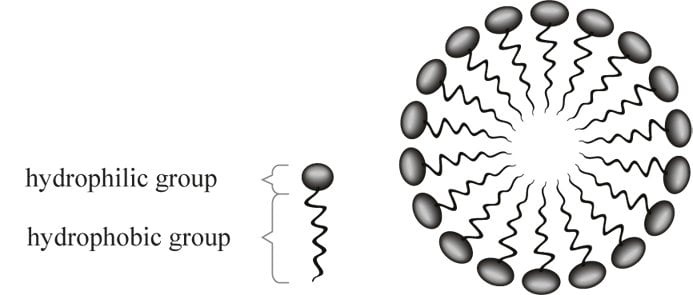 When the proteins start uncoiling and begin coming together, you have the formation of a fluffy meringue mix.
When the proteins start uncoiling and begin coming together, you have the formation of a fluffy meringue mix.

So how do we know that this fluffy mixture won’t eventually go flat? Quite simply, the surface of the air bubbles is crowded with these proteins. Water molecules are attracted to each other, but these proteins force them apart, which causes them to spread.
Some of the proteins form a stable network, which keeps the air bubbles from popping.
When you put your meringue mix in the oven, another protein in the egg (ova-albumin) causes the meringue to harden. After a short stint in the oven, you have your finished product. Delicious!
Mix ‘Em Up!
I have a perfect potato dish in mind for dinner, but I need a sauce to go with it. I’m going to try my hand at making a Hollandaise sauce. Let’s see what an egg has to go through to make that perfect topping.
As you know, oil and water don’t mix. They’re immisicble liquids.
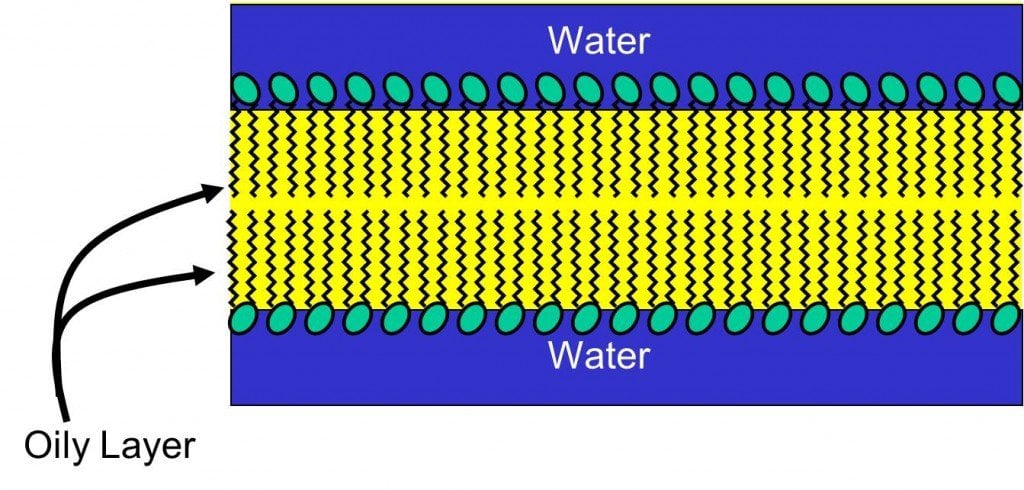
The reason that matters is that Hollandaise sauce requires the blending of oil with the protein-water mix. Uh-oh… what now? Not to worry, this is when the egg yolk comes to the rescue! Apparently, egg yolks act as emulsifiers.
Now, there’s no use in stirring oil and water together. They may mix for a while, but they’ll eventually separate back into their more comfortable states. Therefore, you need to get the oil, egg yolk, and all the seasonings of the sauce ready to go.
Starting off with low heat – just enough to get those proteins all uncoiled and ready for bonding – begin to whisk. Whisk whisk whisk until your arm starts getting tired.
What will happen is that the water-loving end of the emulsifier will stick to the water, while the water-hating end of it will stick to the oil.
This dual set of partnerships will stop the oil droplets from coming together.
Mmmmm… your sauce is coming together beautifully, isn’t it?
Now it’s time to add the butter. Make sure it’s melted, since that allows the emulsifier to bind to it more easily. Using hard butter will just make it harder to work with.

I seriously had no idea that making egg dishes and sauces was about far more than simply following instructions in a recipe and adding one thing after the other. There is so much more to cooking eggs – and many other dishes too!
Knowing why we add melted butter or why we keep the heat on low is fascinating, and not just for chefs. Knowing the science behind our favorite foods makes the process of cooking even more fun.
Oh, and don’t worry… cooking an egg by heating it, whipping it, or frying it won’t change the nutritional content one bit.
These cooked varieties still have the same amount of protein as a raw egg, but the things you add to it can have a major impact. Do you want extra cheese and butter in those eggs? Sure, that’s one express ticket to Cholesterol-ville.
I’ve never had so much appreciation for an egg. Also, I’ve never had such a serious craving for an omelet!













